-

 Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia.
Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia. -

 Enabling Quick Intubations Specially designed to work with GlideScope® video laryngoscopes, GlideRite® Stylets enable efficient endotracheal tube placement. GlideRite® Single-use Stylet - Small Designed for use in ET tubes 3.0 mm to 4.0 mm GlideRite® Single-use Stylet - Large Designed for use in ET tubes 6.0 mm and larger GlideRite® Rigid Stylet Designed for use in ET tubes 6.0 mm and larger GlideRite® DLT Stylet Designed for use in double lumen ET tubes 6.0 mm and larger
Enabling Quick Intubations Specially designed to work with GlideScope® video laryngoscopes, GlideRite® Stylets enable efficient endotracheal tube placement. GlideRite® Single-use Stylet - Small Designed for use in ET tubes 3.0 mm to 4.0 mm GlideRite® Single-use Stylet - Large Designed for use in ET tubes 6.0 mm and larger GlideRite® Rigid Stylet Designed for use in ET tubes 6.0 mm and larger GlideRite® DLT Stylet Designed for use in double lumen ET tubes 6.0 mm and larger -

 Real-time Airway View The GlideScope® AVL video laryngoscope system delivers a consistently clear, real-time view of the airway. Get a better airway view with a large, full colour monitor. Improve training and document intubation results with features such as snapshot and video recording. 6 Single-use sizes available. The AVL system offers 6 single-use GVL Stat sizes for patients from neonate to large adult.
Real-time Airway View The GlideScope® AVL video laryngoscope system delivers a consistently clear, real-time view of the airway. Get a better airway view with a large, full colour monitor. Improve training and document intubation results with features such as snapshot and video recording. 6 Single-use sizes available. The AVL system offers 6 single-use GVL Stat sizes for patients from neonate to large adult. -

 A Brighter Vision in Single-use Bronchoscopy GlideScope® Bflex™ is a single-use bronchoscope designed with exceptional image quality and manoeuvrability for bronchoscopy procedures and difficult airways. BFlex single-use bronchoscopes combine the convenience and accessibility of single-use tools with the performance you expect from a reusable bronchoscope. IMPROVE EFFICIENCY WHILE REDUCING COSTS Single-use bronchoscopes may provide better accessibility and cost effectiveness compared to reusable bronchoscopes.
A Brighter Vision in Single-use Bronchoscopy GlideScope® Bflex™ is a single-use bronchoscope designed with exceptional image quality and manoeuvrability for bronchoscopy procedures and difficult airways. BFlex single-use bronchoscopes combine the convenience and accessibility of single-use tools with the performance you expect from a reusable bronchoscope. IMPROVE EFFICIENCY WHILE REDUCING COSTS Single-use bronchoscopes may provide better accessibility and cost effectiveness compared to reusable bronchoscopes. -

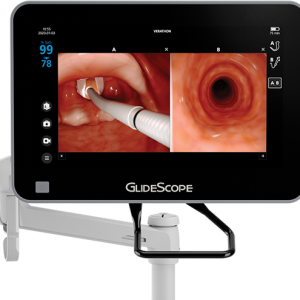 The power to see more GlideScope® Core™, the most comprehensive and flexible airway visualization system for video laryngoscopy, bronchoscopy and multimodal airway procedures. GlideScope Core is an all-in-one system offering immediate access to the tools you need to visualize the airway and tracheobronchial tree. Designed around a high-definition, touchscreen monitor and comprehensive workstation, GlideScope Core delivers elevated visibility and improved workflow. GlideScope Core is the first airway visualization system to offer live multimodal image capability, giving you the power to view the airway via a GlideScope video laryngoscope and BFlex™ single-use flexible bronchoscope – simultaneously. Choose a 10” OR 15” Touchscreen monitor.
The power to see more GlideScope® Core™, the most comprehensive and flexible airway visualization system for video laryngoscopy, bronchoscopy and multimodal airway procedures. GlideScope Core is an all-in-one system offering immediate access to the tools you need to visualize the airway and tracheobronchial tree. Designed around a high-definition, touchscreen monitor and comprehensive workstation, GlideScope Core delivers elevated visibility and improved workflow. GlideScope Core is the first airway visualization system to offer live multimodal image capability, giving you the power to view the airway via a GlideScope video laryngoscope and BFlex™ single-use flexible bronchoscope – simultaneously. Choose a 10” OR 15” Touchscreen monitor. -

 Handheld System - Portability without Compromise Verathon’s second generation handheld system delivering the confidence, clarity and ease-of-use of GlideScope®, your trusted brand of video laryngoscopes designed for rapid, first pass success1. GlideScope Go 2 is IEC 60601-1-12:2014 certified for use in Emergency Medical Services (EMS) environments. Get the confidence of GlideScope® whenever and wherever you need it, with the portable GlideScope® Go™ — the new handheld system created for both hospital and pre-hospital settings. Intubate routine and difficult airways with the handheld system, designed for 1st pass success.
Handheld System - Portability without Compromise Verathon’s second generation handheld system delivering the confidence, clarity and ease-of-use of GlideScope®, your trusted brand of video laryngoscopes designed for rapid, first pass success1. GlideScope Go 2 is IEC 60601-1-12:2014 certified for use in Emergency Medical Services (EMS) environments. Get the confidence of GlideScope® whenever and wherever you need it, with the portable GlideScope® Go™ — the new handheld system created for both hospital and pre-hospital settings. Intubate routine and difficult airways with the handheld system, designed for 1st pass success.1. Silverberg, Michael J., et al. “Comparison of Video Laryngoscopy Versus Direct Laryngoscopy During Urgent Endotracheal Intubation: A Randomized Controlled Trial.” Critical Care Medicine 43.3 (2014): 636-641.
-

 Designed for 1st Pass Success GlideScope® Spectrum™ Single-use video laryngoscopes combine fully disposable low profile blades with the latest advancements in lighting and camera technology. Compatible with the GlideScope® Go, GlideScope® Core and GlideScope® Video Monitor. With the Spectrum system, seven blade sizes enable quick intubations across a wide range of patient types, weights, and clinical settings. The LoPro blades offer the signature GlideScope blade angulation and cover a patient range from neonate to large adult. The DirectView MAC blades extend system flexibility, incorporating the look and feel of traditional MAC blades with the confidence of a GlideScope. Available in 7 sizes.
Designed for 1st Pass Success GlideScope® Spectrum™ Single-use video laryngoscopes combine fully disposable low profile blades with the latest advancements in lighting and camera technology. Compatible with the GlideScope® Go, GlideScope® Core and GlideScope® Video Monitor. With the Spectrum system, seven blade sizes enable quick intubations across a wide range of patient types, weights, and clinical settings. The LoPro blades offer the signature GlideScope blade angulation and cover a patient range from neonate to large adult. The DirectView MAC blades extend system flexibility, incorporating the look and feel of traditional MAC blades with the confidence of a GlideScope. Available in 7 sizes. -

 Lightweight titanium for added strength and durability Innovative construction and blade styles for a wide array of clinical demands. Incorporating an advanced digital camera with an integrated LED light source, GlideScope® Titanium™ laryngoscopes connect to the all new GlideScope® Core™ system for real time viewing and recording. and with a choice of low-profile blades in two distinct options, you can choose the way you want to work. Available in 5 sizes.
Lightweight titanium for added strength and durability Innovative construction and blade styles for a wide array of clinical demands. Incorporating an advanced digital camera with an integrated LED light source, GlideScope® Titanium™ laryngoscopes connect to the all new GlideScope® Core™ system for real time viewing and recording. and with a choice of low-profile blades in two distinct options, you can choose the way you want to work. Available in 5 sizes. -

 Reusable Video Baton GlideScope® Video Baton 2.0 Large is a reusable video baton that works with single-use stats to provide a consistently clear, real-time view of the airway. We are continually developing new devices and accessories to meet the demands of your changing environment. Our Video Baton 2.0 Large is compatible with GlideScope® Go™ and GlideScope® Core™, making it easier to use and to purchase products from a trusted single source.
Reusable Video Baton GlideScope® Video Baton 2.0 Large is a reusable video baton that works with single-use stats to provide a consistently clear, real-time view of the airway. We are continually developing new devices and accessories to meet the demands of your changing environment. Our Video Baton 2.0 Large is compatible with GlideScope® Go™ and GlideScope® Core™, making it easier to use and to purchase products from a trusted single source. -

 Genetically modified crops cover over 10% of global agricultural land. More than one hundred different genetically modified plants are grown on over 160 million hectares in more than 29 countries. The main producing countries are the USA, Brazil, Argentina, Canada and India. Soybeans are the most highly cultivated genetically modified plant, followed by corn, cotton and canola. According to EU Regulation 1829/2003 and 1830/2003, GMO content of more than 0.9% in foods and ingredients must be labeled. Other countries and regions have similar rules with similar limits. For reliable analysis of GMOs in food and animal feed, BIOTECON Diagnostics offers a broad range of solutions.
Genetically modified crops cover over 10% of global agricultural land. More than one hundred different genetically modified plants are grown on over 160 million hectares in more than 29 countries. The main producing countries are the USA, Brazil, Argentina, Canada and India. Soybeans are the most highly cultivated genetically modified plant, followed by corn, cotton and canola. According to EU Regulation 1829/2003 and 1830/2003, GMO content of more than 0.9% in foods and ingredients must be labeled. Other countries and regions have similar rules with similar limits. For reliable analysis of GMOs in food and animal feed, BIOTECON Diagnostics offers a broad range of solutions. -

 AMT Balloon G-Tube The AMT Balloon G-Tube is a Silicone replacement gastrostomy tube (G-Tube). The G-Tube is placed through the abdominal wall to give direct access to the stomach and has an adjustable external bolster minimising surface contact with the skin, allowing the tissue to breathe and can be adjusted up to 10cm in length.
AMT Balloon G-Tube The AMT Balloon G-Tube is a Silicone replacement gastrostomy tube (G-Tube). The G-Tube is placed through the abdominal wall to give direct access to the stomach and has an adjustable external bolster minimising surface contact with the skin, allowing the tissue to breathe and can be adjusted up to 10cm in length. -

 Internal Quality Control Software from Technopath Manufacturing Ltd. IAMQC Daily is a comprehensive Internal Quality Control software that applies Westgard and/or any user-defined QC rules to individual QC results. The software automatically builds interactive Levey-Jennings charts and tables and provides summary and detailed customised reports to the end user. IAMQC Daily integrates with Microsoft Excel to produce customised electronic reports. The system also allows the import of pre-defined templates, resulting in instant system setups. Users can create audit trails, action logs and summary reports at the click of a button. IAMQC Daily comprises a centralised program that facilitates the analysis of multiple QC materials, across numerous departments in a laboratory setting. The system can also be integrated with IAMQC Peer to satisfy both Internal and External QC requirements. IAMQC Daily offers a centralised review of all QC data from all laboratories/instruments. Central administrator access facilitates managers to review QC performance at multiple facilities – no need to visit each laboratory site. Closer monitoring of QC from remote locations without additional costs provides a flexible option for managing the inter and intra-lab performance. The software works on an ‘open’ platform that allows the end user to add all types of control material from a range of laboratories. Both IAMQC Daily and Expert can run using an internet OR intranet connection. Each PC license allows the user to manage an unlimited number of departments, control materials (not limited to Technopath materials) and instruments. An unlimited number of user logins can be added to the system at any stage. An administrator module can also manage user logins, customising the functionality that is available to each user. All data can also be filtered using the same logic to apply a user-friendly atmosphere and save time scrolling through data.
Internal Quality Control Software from Technopath Manufacturing Ltd. IAMQC Daily is a comprehensive Internal Quality Control software that applies Westgard and/or any user-defined QC rules to individual QC results. The software automatically builds interactive Levey-Jennings charts and tables and provides summary and detailed customised reports to the end user. IAMQC Daily integrates with Microsoft Excel to produce customised electronic reports. The system also allows the import of pre-defined templates, resulting in instant system setups. Users can create audit trails, action logs and summary reports at the click of a button. IAMQC Daily comprises a centralised program that facilitates the analysis of multiple QC materials, across numerous departments in a laboratory setting. The system can also be integrated with IAMQC Peer to satisfy both Internal and External QC requirements. IAMQC Daily offers a centralised review of all QC data from all laboratories/instruments. Central administrator access facilitates managers to review QC performance at multiple facilities – no need to visit each laboratory site. Closer monitoring of QC from remote locations without additional costs provides a flexible option for managing the inter and intra-lab performance. The software works on an ‘open’ platform that allows the end user to add all types of control material from a range of laboratories. Both IAMQC Daily and Expert can run using an internet OR intranet connection. Each PC license allows the user to manage an unlimited number of departments, control materials (not limited to Technopath materials) and instruments. An unlimited number of user logins can be added to the system at any stage. An administrator module can also manage user logins, customising the functionality that is available to each user. All data can also be filtered using the same logic to apply a user-friendly atmosphere and save time scrolling through data. -

 Select QC Rules and make meaningful QC decisions IAMQC Expert, from Technopath Manufacturing Ltd, is an interactive system that helps front line laboratory staff select QC rules, reduce unnecessary repeats and make meaningful QC decisions. IAMQC Expert allows the end-user to monitor method performance relative to clinical requirements and focus on the tests that require their attention. Times and technologies are changing rapidly. Instruments and methodologies are more accurate, precise and stable than they were a decade ago. Most laboratories have adopted these new technical advances, but few have modified their QC processes to match. Many laboratories are still using a 1-2s rule as recommended by Levey and Jennings in 1951. Technopath Manufacturing Ltd have designed a QC system that will alert users to significant changes and not generate QC flags when the system is operating safely within acceptable limits. The system compares method performance to defined quality requirements (rather than to last month’s data) and recommends QC strategies that will warn users when QC data points exceed acceptable performance - with a minimal number of false flags. In the design of our QC system we “balance” the quality control system to meet the changing performance and stability of the analytical system.
Select QC Rules and make meaningful QC decisions IAMQC Expert, from Technopath Manufacturing Ltd, is an interactive system that helps front line laboratory staff select QC rules, reduce unnecessary repeats and make meaningful QC decisions. IAMQC Expert allows the end-user to monitor method performance relative to clinical requirements and focus on the tests that require their attention. Times and technologies are changing rapidly. Instruments and methodologies are more accurate, precise and stable than they were a decade ago. Most laboratories have adopted these new technical advances, but few have modified their QC processes to match. Many laboratories are still using a 1-2s rule as recommended by Levey and Jennings in 1951. Technopath Manufacturing Ltd have designed a QC system that will alert users to significant changes and not generate QC flags when the system is operating safely within acceptable limits. The system compares method performance to defined quality requirements (rather than to last month’s data) and recommends QC strategies that will warn users when QC data points exceed acceptable performance - with a minimal number of false flags. In the design of our QC system we “balance” the quality control system to meet the changing performance and stability of the analytical system. -

 Innovative, real-time, Peer Comparison Software from Technopath Manufacturing Ltd. The web based system facilitates laboratories testing the same lot number of control material to access valuable information from their colleagues through peer comparison. The reports that are generated in IAMQC Peer compare the accuracy and precision of analytical processes between laboratories and peer groups. This information can be extremely valuable, indicating the user’s performance relative to their peer group and also providing powerful troubleshooting tools when attempting to resolve potential problems. To participate in IAMQC Peer, each individual laboratory submits their individual results or summary statistics (mean, standard deviation, and number of data points) to the central database maintained by Technopath Manufacturing Ltd. Laboratories data may be submitted manually on-line or, alternatively, captured by one of our many interfacing options. The information provided by IAMQC Peer can be used on a monthly basis to evaluate how well lab’s methods are operating, relative to the overall peer group. Users can also look at this peer data in real-time, interactive, tables online, when they are investigating a potential problem with accuracy or precision for an individual method.
Innovative, real-time, Peer Comparison Software from Technopath Manufacturing Ltd. The web based system facilitates laboratories testing the same lot number of control material to access valuable information from their colleagues through peer comparison. The reports that are generated in IAMQC Peer compare the accuracy and precision of analytical processes between laboratories and peer groups. This information can be extremely valuable, indicating the user’s performance relative to their peer group and also providing powerful troubleshooting tools when attempting to resolve potential problems. To participate in IAMQC Peer, each individual laboratory submits their individual results or summary statistics (mean, standard deviation, and number of data points) to the central database maintained by Technopath Manufacturing Ltd. Laboratories data may be submitted manually on-line or, alternatively, captured by one of our many interfacing options. The information provided by IAMQC Peer can be used on a monthly basis to evaluate how well lab’s methods are operating, relative to the overall peer group. Users can also look at this peer data in real-time, interactive, tables online, when they are investigating a potential problem with accuracy or precision for an individual method. -

 Detects more beta-lactam residues including cephalexin at or below established maximum residue limits.
Detects more beta-lactam residues including cephalexin at or below established maximum residue limits.- Test cold milk right from the truck, refrigerator or bulk tank
- Robust and flexible
- Cow, Goat, Sheep, Mare, Buffalo, Camel
- Variety of milk types (Normal, UHT, Sterilised, reconstituted milk powder, thawed, skimmed)
- Read results after 6 minutes
- ILVO validated
- Lowest rate of false positives
-

 The following additional IDEXX SNAP Tests are available and validated to detect residues in raw, commingled cow milk. SNAP Tetracycline Test Tetracycline is one of the most commonly used antibiotics—make sure your milk is within residue limits. SNAP Gentamicin Test Ensures that residues from this common mastitis antibiotic are within regulatory limits. SNAP Aflatoxin M1 Test AFM1 is a potent carcinogen that can enter the milk supply if cows consume contaminated feed. SNAP Sulfamethazine Test Detects sulfamethazine residues at or below the safe/tolerance level. A heater block is used during the heat/incubation step.
The following additional IDEXX SNAP Tests are available and validated to detect residues in raw, commingled cow milk. SNAP Tetracycline Test Tetracycline is one of the most commonly used antibiotics—make sure your milk is within residue limits. SNAP Gentamicin Test Ensures that residues from this common mastitis antibiotic are within regulatory limits. SNAP Aflatoxin M1 Test AFM1 is a potent carcinogen that can enter the milk supply if cows consume contaminated feed. SNAP Sulfamethazine Test Detects sulfamethazine residues at or below the safe/tolerance level. A heater block is used during the heat/incubation step. Description: Use with all IDEXX SNAP tests that include a heat/incubation step.
Ensures correct, even temperatures for reliable, consistent results.
May be used in conjunction with the SNAPshot DSR Reader to read and display test results.
Description: Use with all IDEXX SNAP tests that include a heat/incubation step.
Ensures correct, even temperatures for reliable, consistent results.
May be used in conjunction with the SNAPshot DSR Reader to read and display test results. -

 Detects more beta-lactam and tetracycline residues including cephalexin at or below established maximum residue limits.
Detects more beta-lactam and tetracycline residues including cephalexin at or below established maximum residue limits.- Test cold milk right from the truck, refrigerator or bulk tank
- Robust and flexible
- Cow, Goat, Sheep, Mare, Buffalo, Camel
- Variety of milk types (Normal, UHT, Sterilised, reconstituted milk powder, thawed, skimmed)
- Read results after 6 minutes
- ILVO validated
- Lowest rate of false positives
 Description: Use with all IDEXX SNAP tests that include a heat/incubation step.
Ensures correct, even temperatures for reliable, consistent results.
May be used in conjunction with the SNAPshot DSR Reader to read and display test results.
Description: Use with all IDEXX SNAP tests that include a heat/incubation step.
Ensures correct, even temperatures for reliable, consistent results.
May be used in conjunction with the SNAPshot DSR Reader to read and display test results. -

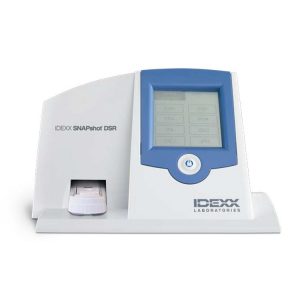 Quickly reads and displays test results Enter data quickly Touch-screen display with numeric data entry, or full alphanumeric data entry with SNAPconnect software or an attached keyboard. Get a clearer picture Upgraded vision system and display make results easy to read. Choose your language Use the SNAPshot DSR display in any of these languages: English, French, German, Spanish, Italian, Portuguese, Chinese, Japanese
Quickly reads and displays test results Enter data quickly Touch-screen display with numeric data entry, or full alphanumeric data entry with SNAPconnect software or an attached keyboard. Get a clearer picture Upgraded vision system and display make results easy to read. Choose your language Use the SNAPshot DSR display in any of these languages: English, French, German, Spanish, Italian, Portuguese, Chinese, Japanese -

 Precision. Confidence. Compliance. Technopath is proud to partner with Diagast, a global leader in immunohaematology, to deliver a comprehensive range of quality control solutions designed to ensure accuracy, reliability, and full regulatory compliance in transfusion laboratories with ISO 15189 requirements. Diagast’s immunohaematology quality control portfolio supports laboratories in maintaining the highest standards across blood grouping, antibody screening, crossmatching, and reagent performance verification—helping safeguard patient outcomes at every stage of the transfusion pathway. Comprehensive Quality Control for Transfusion Safety In immunohaematology, precision is critical. Diagast’s quality controls are specifically developed to:
Precision. Confidence. Compliance. Technopath is proud to partner with Diagast, a global leader in immunohaematology, to deliver a comprehensive range of quality control solutions designed to ensure accuracy, reliability, and full regulatory compliance in transfusion laboratories with ISO 15189 requirements. Diagast’s immunohaematology quality control portfolio supports laboratories in maintaining the highest standards across blood grouping, antibody screening, crossmatching, and reagent performance verification—helping safeguard patient outcomes at every stage of the transfusion pathway. Comprehensive Quality Control for Transfusion Safety In immunohaematology, precision is critical. Diagast’s quality controls are specifically developed to:- Monitor assay performance across routine and specialised testing
- Verify reagent integrity and system functionality
- Ensure compliance with ISO 15189 and local regulatory standards (HPRA, UKAS)
- Reduce risk of transfusion errors and improve laboratory confidence
-

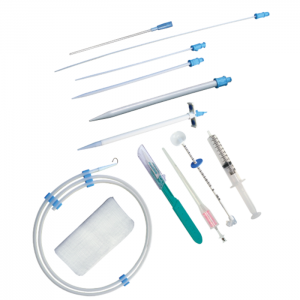 Comprehensive Kit The Initial Placement Kit reduces the trauma associated with stoma tract development. Use laparoscopically, endoscopically or with fluoroscopic guidance. Eliminates a secondary procedure to replace with a low profile. One endoscopic pass reducing trauma. For use with 12-18FR devices.
Comprehensive Kit The Initial Placement Kit reduces the trauma associated with stoma tract development. Use laparoscopically, endoscopically or with fluoroscopic guidance. Eliminates a secondary procedure to replace with a low profile. One endoscopic pass reducing trauma. For use with 12-18FR devices. -

 Nutricare® ISOSAF Nutricare ISOSAF is a fully radiopaque polyurethane tube with clear numbered cm markings. It is fitted with the new ENFit (ISO 80369-3) connector. Each pack includes the NG tube with guidewire*, fixation plaster, record of insertion sticker and safety literature. NJ tube clearly identifiable with blue cap & tube markings. * 14 & 16Fr Supplied without guidewire & safety literature
Nutricare® ISOSAF Nutricare ISOSAF is a fully radiopaque polyurethane tube with clear numbered cm markings. It is fitted with the new ENFit (ISO 80369-3) connector. Each pack includes the NG tube with guidewire*, fixation plaster, record of insertion sticker and safety literature. NJ tube clearly identifiable with blue cap & tube markings. * 14 & 16Fr Supplied without guidewire & safety literature -

 Introducing Nutricare® Infant Clear Nutricare Infant Clear from GBUK Group is a polyurethane tube with clear numbered cm marking. It is fitted with the new ENFit (ISO 80369-3) connector. Each single blister includes the NG tube and fixation plaster. *Blue stripe on tube is for illustrative purposes only
Introducing Nutricare® Infant Clear Nutricare Infant Clear from GBUK Group is a polyurethane tube with clear numbered cm marking. It is fitted with the new ENFit (ISO 80369-3) connector. Each single blister includes the NG tube and fixation plaster. *Blue stripe on tube is for illustrative purposes only -

 AMT MiniONE® Capsule Button The AMT MiniONE® Non-Balloon Button lies flush against the skin of the abdomen and has a small internal bolster that sits inside the stomach to prevent the device from falling out. The MiniONE® Capsule Non-Balloon Button combines the advantages of a low-profile device with the longer life of a non-balloon device.
AMT MiniONE® Capsule Button The AMT MiniONE® Non-Balloon Button lies flush against the skin of the abdomen and has a small internal bolster that sits inside the stomach to prevent the device from falling out. The MiniONE® Capsule Non-Balloon Button combines the advantages of a low-profile device with the longer life of a non-balloon device. -

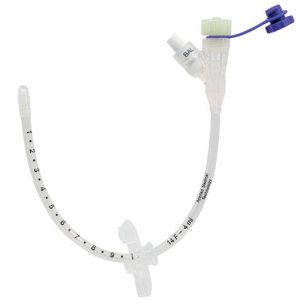
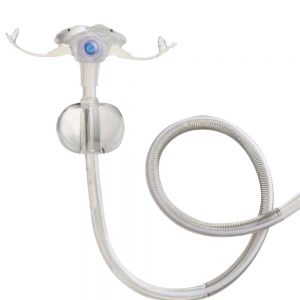 The AMT G-Jet The AMT G-Jet is a low profile Gastric-Jejunal enteral feeding tube which is used to help patients meet their nutritional needs by feeding directly into the small bowel whilst being able to vent the stomach at the same time. Just like the AMT’s Mini Compact Balloon Buttons, the G-JET Button’s balloon is shaped to cover more surface area and minimise leaks at the stoma site. It seals better to reduce leaks that can lead to gastric ulceration. Gastric holes are located immediately after balloon to ensure gastric-only access. Co-extruded radiopaque stripes provide clear radiographic identification. Mutually-exclusive ports. Ports and extension sets are colour-coded for visual differentiation. Available in 14Fr and 16Fr.
The AMT G-Jet The AMT G-Jet is a low profile Gastric-Jejunal enteral feeding tube which is used to help patients meet their nutritional needs by feeding directly into the small bowel whilst being able to vent the stomach at the same time. Just like the AMT’s Mini Compact Balloon Buttons, the G-JET Button’s balloon is shaped to cover more surface area and minimise leaks at the stoma site. It seals better to reduce leaks that can lead to gastric ulceration. Gastric holes are located immediately after balloon to ensure gastric-only access. Co-extruded radiopaque stripes provide clear radiographic identification. Mutually-exclusive ports. Ports and extension sets are colour-coded for visual differentiation. Available in 14Fr and 16Fr. -

 AMT Mini and MiniONE® Balloon Button Range AMT’s Mini and MiniONE® family of low profile balloon G-Tubes are designed to reduce leaks, improve comfort, and improve patient outcomes. These devices sit flush against the skin and can be concealed under garments easily allowing the patient a wide range of motion. The buttons can be purchased as single units: MiniONE® Balloon Button SM1-5 A single MiniONE Balloon Button with 5ml syringe & introducer. Mini Balloon Button E5 A single MINI Balloon Button E5 with 5ml syringe.
AMT Mini and MiniONE® Balloon Button Range AMT’s Mini and MiniONE® family of low profile balloon G-Tubes are designed to reduce leaks, improve comfort, and improve patient outcomes. These devices sit flush against the skin and can be concealed under garments easily allowing the patient a wide range of motion. The buttons can be purchased as single units: MiniONE® Balloon Button SM1-5 A single MiniONE Balloon Button with 5ml syringe & introducer. Mini Balloon Button E5 A single MINI Balloon Button E5 with 5ml syringe. -

 Completely adaptable system for all applications The LUXOmatic from LMB is a premium blood separator for blood products of the highest quality. The dual press system allows for blood separation in two places at the same time.
Completely adaptable system for all applications The LUXOmatic from LMB is a premium blood separator for blood products of the highest quality. The dual press system allows for blood separation in two places at the same time.- Quick blood separation bags and high recovery of thrombocytes
- Precise treatment of all Top/Top and Top/Bottom processes
- CRYO application
- Thrombocytes from Buffy Coat and PRP
- Adaptive optical sensor system for best possible distinction between red cells, thrombocytes and plasma
- Precise and durable mechanical system
-

 Space saver and adaptable system for individual applications The LUXOmatic V2 from LMB is a premium blood separator with motor driven plates, touch screen and integrated bar code scanner.
Space saver and adaptable system for individual applications The LUXOmatic V2 from LMB is a premium blood separator with motor driven plates, touch screen and integrated bar code scanner.- Sophisticated motor driven blood separator
- Two independent separation places in one device – Saves working space, time and provides best economic benefit
- High quality of separated blood products
- Heavy duty industrial scale separations
- Paediatric bags extraction possibility
- Ergonomic design
-

 Phenotype MicroArrays for Mammalian Cells Using Biolog’s Phenotype MicroArray™ Technology, any type of cell can be characterised for metabolically related phenotypes in over 1,000 assays in a simple, rapid, and cost-effective manner. By measuring a cell’s metabolic activity, including responses to a genetic alteration or chemical stimuli, this integrated system of cellular assays, instrumentation, and bioinformatic software reveals unique and insightful information on metabolic pathway activities, and cellular sensitivity to nutrients, hormones, cytokines, anti-cancer agents, and ions. By measuring energy output of cells, the technology also provides new assays for understanding mitochondrial function and toxicity. Phenotype MicroArrays enable insight and discovery to expedite scientific publications. This is accomplished using 96 well microplates, pre-loaded with hundreds of KEGG pathway-matched metabolic substrates, and metabolic effectors. Phenotype MicroArrays are a proven method of cellular screening that is beneficial in a wide range of research applications:
Phenotype MicroArrays for Mammalian Cells Using Biolog’s Phenotype MicroArray™ Technology, any type of cell can be characterised for metabolically related phenotypes in over 1,000 assays in a simple, rapid, and cost-effective manner. By measuring a cell’s metabolic activity, including responses to a genetic alteration or chemical stimuli, this integrated system of cellular assays, instrumentation, and bioinformatic software reveals unique and insightful information on metabolic pathway activities, and cellular sensitivity to nutrients, hormones, cytokines, anti-cancer agents, and ions. By measuring energy output of cells, the technology also provides new assays for understanding mitochondrial function and toxicity. Phenotype MicroArrays enable insight and discovery to expedite scientific publications. This is accomplished using 96 well microplates, pre-loaded with hundreds of KEGG pathway-matched metabolic substrates, and metabolic effectors. Phenotype MicroArrays are a proven method of cellular screening that is beneficial in a wide range of research applications:- Genotype/phenotype studies
- Cell line characterisation and QC
- Cellular metabolism, metabolic disorders, nutrition
- Cell energetics growth and death
- Hormone effects on cells
- Metabolic reprogramming in cancer, Warburg effect, anti-cancer drug sensitivity
- Mitochondrial toxicology in chemical/drug screens
- Stem cells and differentiation
- Cell line and bio process development
-

 Analysing Mitochondria at Higher Resolution with MitoPlate™ Technology Analysing Mitochondria at higher resolution with Biolog Technology MitoPlates™ provide a powerful new research tool by allowing scientists to run preconfigured sets of 96 mitochondrial function assays in one experiment. Mitochondria can be interrogated and characterised in novel ways, looking at rates of substrate metabolism, sensitivity to drugs and other chemicals, and effects of mutations in mitochondria-related genes. Assay Principle Mitochondrial function is assayed by measuring the rates of electron flow into and through the electron transport chain from metabolic substrates that produce NAD(P)H or FADH2 such as L-malate, succinate, pyruvate, etc. Each substrate follows a different route, using different transporters to enter the mitochondria and different dehydrogenases to produce NAD(P)H or FADH2, The electrons travel from the beginning (complex 1 or 2) to the distal portion of the electron transport chain where a tetrazolium redox dye (MC) acts as a terminal electron acceptor that turns purple upon reduction. Additional MitoPlate assays probe the sensitivity of the mitochondria to a set of 22 diverse inhibitors. Investigate how mitochondria change with:
Analysing Mitochondria at Higher Resolution with MitoPlate™ Technology Analysing Mitochondria at higher resolution with Biolog Technology MitoPlates™ provide a powerful new research tool by allowing scientists to run preconfigured sets of 96 mitochondrial function assays in one experiment. Mitochondria can be interrogated and characterised in novel ways, looking at rates of substrate metabolism, sensitivity to drugs and other chemicals, and effects of mutations in mitochondria-related genes. Assay Principle Mitochondrial function is assayed by measuring the rates of electron flow into and through the electron transport chain from metabolic substrates that produce NAD(P)H or FADH2 such as L-malate, succinate, pyruvate, etc. Each substrate follows a different route, using different transporters to enter the mitochondria and different dehydrogenases to produce NAD(P)H or FADH2, The electrons travel from the beginning (complex 1 or 2) to the distal portion of the electron transport chain where a tetrazolium redox dye (MC) acts as a terminal electron acceptor that turns purple upon reduction. Additional MitoPlate assays probe the sensitivity of the mitochondria to a set of 22 diverse inhibitors. Investigate how mitochondria change with:- Cell differentiation
- Cancer & Aging
- Neurological disorders
- Metabolic disorders
- Immune cell activation
- Bacterial/viral infection
- Inborn genetic defects
-

 Enteral Medicine Straw Male ENFit connector (ISO 80369-3). The ENFit medicine straw from Enteral UK allows for more accuracy when drawing up feed/medication into your ENFit Enteral Syringe. Available in sterile and non sterile.
Enteral Medicine Straw Male ENFit connector (ISO 80369-3). The ENFit medicine straw from Enteral UK allows for more accuracy when drawing up feed/medication into your ENFit Enteral Syringe. Available in sterile and non sterile. -

 Phenotype MicroArrays for Microbial Cells Biolog's Phenotype MicroArray™ Technology enables researchers to characterise cells in up to 1,920 assays and evaluate cell changes under thousands of culture conditions and physiological states in a simple, rapid, efficient and cost-effective manner. By measuring a cell’s response to a genetic or environmental alteration, this integrated system of cellular assays, instrumentation, and bioinformatics software reveals invaluable information to speed insight and discovery and expedite scientific publication. Phenotype MicroArrays (PMs) have broad applicability for genotype-phenotype characterisation as well as for determining optimal conditions for cellular growth, sporulation and germination, production of secondary metabolites, or enzymatic activities in cell lines. Phenotype MicroArrays are a proven method of cellular screening that is extremely beneficial in a wide range of research applications:
Phenotype MicroArrays for Microbial Cells Biolog's Phenotype MicroArray™ Technology enables researchers to characterise cells in up to 1,920 assays and evaluate cell changes under thousands of culture conditions and physiological states in a simple, rapid, efficient and cost-effective manner. By measuring a cell’s response to a genetic or environmental alteration, this integrated system of cellular assays, instrumentation, and bioinformatics software reveals invaluable information to speed insight and discovery and expedite scientific publication. Phenotype MicroArrays (PMs) have broad applicability for genotype-phenotype characterisation as well as for determining optimal conditions for cellular growth, sporulation and germination, production of secondary metabolites, or enzymatic activities in cell lines. Phenotype MicroArrays are a proven method of cellular screening that is extremely beneficial in a wide range of research applications:- Discovering effects of loss or gain of gene function.
- Measuring/documenting changes in cell metabolism over time or under different environmental conditions.
- Improvement and QC of phenotypic stability of cell lines.
- Improved efficacy in the production of compounds in biological fermentation processes.
- Evaluation of new drug/antibiotic candidates in toxicological profiling and mode of action studies.
-

 Microbial ID microplates from Biolog Biolog Solutions for Microbial Identification Rapidly and accurately identify nearly 3,000 species of aerobic and anaerobic bacteria, yeast, and fungi. In addition to species-level identification, Biolog’s advanced phenotypic technology provides valuable information on the functional properties of strains. Biolog’s carbon source utilisation technology identifies environmental and pathogenic microorganisms by producing a characteristic pattern or “metabolic fingerprint” from discrete test reactions performed within a 96-well microplate. Culture suspensions are tested with a panel of pre-selected assays, then incubated, read and compared to our extensive database. Biolog’s Microbial ID microplates incorporate a patented redox tetrazolium dye that changes colour as a result of cellular respiration, providing a metabolic fingerprint. Our complete solution includes instruments, consumables, databases, software.
Microbial ID microplates from Biolog Biolog Solutions for Microbial Identification Rapidly and accurately identify nearly 3,000 species of aerobic and anaerobic bacteria, yeast, and fungi. In addition to species-level identification, Biolog’s advanced phenotypic technology provides valuable information on the functional properties of strains. Biolog’s carbon source utilisation technology identifies environmental and pathogenic microorganisms by producing a characteristic pattern or “metabolic fingerprint” from discrete test reactions performed within a 96-well microplate. Culture suspensions are tested with a panel of pre-selected assays, then incubated, read and compared to our extensive database. Biolog’s Microbial ID microplates incorporate a patented redox tetrazolium dye that changes colour as a result of cellular respiration, providing a metabolic fingerprint. Our complete solution includes instruments, consumables, databases, software. -

 Microbial Controls made from your Environmental Isolates Tracking and trending environmental microbial isolates is a growing concern for pharmaceutical, nutraceutical, medical device, personal care product and food manufacturing laboratories. Microbiologics offers a comprehensive program for environmental isolate management. Our team of experts will identify, characterise, preserve and manufacture your strain into a test-ready control format for ongoing QC. Partner with TECHNOPATH and Microbiologics to reduce cost, minimize risk and increase confidence in your environmental monitoring program. Applications & Test Methods
Microbial Controls made from your Environmental Isolates Tracking and trending environmental microbial isolates is a growing concern for pharmaceutical, nutraceutical, medical device, personal care product and food manufacturing laboratories. Microbiologics offers a comprehensive program for environmental isolate management. Our team of experts will identify, characterise, preserve and manufacture your strain into a test-ready control format for ongoing QC. Partner with TECHNOPATH and Microbiologics to reduce cost, minimize risk and increase confidence in your environmental monitoring program. Applications & Test Methods- Antimicrobial Effectiveness Testing - USP <51>
- Aseptic Processing Environment - USP <1116>
- Disinfectant Qualification - USP <116>
- Growth Promotion Testing - USP <61>, <62>, <71>
- Suitability Testing - USP <51>, <61>, <62>, <71>
- Validation of Neutralisation Methods - USP <1227>
- Water for Pharmaceutical Purposes - USP <1231>
-

 Quantitative QC Microorganisms One powerful solution – Many testing applications. Epower, from Microbiologics®, is the embodiment of “flexibility is your friend”. You’ll love the versatility of simply using one product for all of your QC needs – from media QC and presence/absence testing to enumeration testing, water testing, and disinfectant testing. Use a single strain or combine multiple strains for a mixed population. Each quantitative microorganism pellet delivers a specific range of CFU, so you can easily manipulate Epower to deliver virtually any concentration. We know you’ll appreciate the ease and adaptability that Epower offers. Applications & Test Methods
Quantitative QC Microorganisms One powerful solution – Many testing applications. Epower, from Microbiologics®, is the embodiment of “flexibility is your friend”. You’ll love the versatility of simply using one product for all of your QC needs – from media QC and presence/absence testing to enumeration testing, water testing, and disinfectant testing. Use a single strain or combine multiple strains for a mixed population. Each quantitative microorganism pellet delivers a specific range of CFU, so you can easily manipulate Epower to deliver virtually any concentration. We know you’ll appreciate the ease and adaptability that Epower offers. Applications & Test Methods- Microbial detection
- Enumeration
- Verification/validation
- Equipment calibration
- Method validation
- Disinfectant qualification
- Water tests
- Proficiency tests
- Mixed microorganism population challenge
-

 Certified Reference Material The ultimate in authenticity. Epower CRM quantitative quality control microorganisms, from Microbiologics®, are just one passage from the reference strain. If your lab is ISO 17025 accredited, Epower CRM can help you meet the requirements of Section 5.6.3.2 regarding reference materials. This product offers plenty of flexibility, which is a huge benefit in the lab. Each microorganism pellet has a pre-determined range of CFUs, so you can easily manipulate the product to deliver your desired concentration. You can also create a mixed microorganism population by combining multiple Epower CRM strains. As a Certified Reference Material, Epower CRM comes with a comprehensive Certificate of Analysis detailing the strains identity, characteristics and standard deviation. Applications & Test Methods
Certified Reference Material The ultimate in authenticity. Epower CRM quantitative quality control microorganisms, from Microbiologics®, are just one passage from the reference strain. If your lab is ISO 17025 accredited, Epower CRM can help you meet the requirements of Section 5.6.3.2 regarding reference materials. This product offers plenty of flexibility, which is a huge benefit in the lab. Each microorganism pellet has a pre-determined range of CFUs, so you can easily manipulate the product to deliver your desired concentration. You can also create a mixed microorganism population by combining multiple Epower CRM strains. As a Certified Reference Material, Epower CRM comes with a comprehensive Certificate of Analysis detailing the strains identity, characteristics and standard deviation. Applications & Test Methods- ISO 17025 laboratories requiring CRM
- Detection and enumeration methods
- Verification/Validation
- Bioburden determination
- Minimal lethal concentration
- Disinfectant qualification
- Water tests
- Proficiency tests
- Methods requiring a specific CFU range
- Mixed microorganism population challenge
-

 Growth Promotion Testing EZ-Accu Shot, from Microbiologics®, provides a fast, convenient and reliable solution for Growth Promotion Testing, Media Challenge Testing and more. Each instant-dissolve microorganism pellet is designed to deliver 10-100 CFU per inoculum and provides 8 hours of stability after rehydration, so you can meet Pharmacopeia guidelines with ease. Applications & Test Methods
Growth Promotion Testing EZ-Accu Shot, from Microbiologics®, provides a fast, convenient and reliable solution for Growth Promotion Testing, Media Challenge Testing and more. Each instant-dissolve microorganism pellet is designed to deliver 10-100 CFU per inoculum and provides 8 hours of stability after rehydration, so you can meet Pharmacopeia guidelines with ease. Applications & Test Methods- Growth Promotion Testing
- Media Challenge Testing
- Suitability of Counting Methods
- Suitability of Sterility Tests
- Suitability of Tests for Specified Microorganisms
- Microbial Limits Testing
- Microbial Enumeration Testing
- Validation of Neutralization Methods
- Methods requiring a low CFU concentration



