-

 Staphylococcus aureus results in 24 hours 3M™ Petrifilm™ Staph Express Count Plates provide Staphylococcus aureus results. S. aureus appears as distinctive red-violet colonies. Other colonies have to be checked using the Staph express disk. If you encounter high background flora in your staph testing, the 3M™ Petrifilm™ Staph Express Disk can be used to identify Staph aureus from all suspect colonies. The 3M Petrifilm Plate Advantage In today’s environment of heightened focus on food safety and increasingly stringent quality requirements, labs are under pressure to provide tests that are fast, reliable and consistently accurate. 3M Petrifilm Plates have demonstrated consistent performance that has been cited globally with more than 200 certificates, recognitions, validations, and peer-review publications. For more than 30 years, food safety professionals around the world have put their trust in 3M Petrifilm Plates. That’s because 3M Petrifilm Plates have streamlined, standardized and simplified the process of microbial quantitative indicator testing, improving productivity and helping ensure the highest levels of product quality.
Staphylococcus aureus results in 24 hours 3M™ Petrifilm™ Staph Express Count Plates provide Staphylococcus aureus results. S. aureus appears as distinctive red-violet colonies. Other colonies have to be checked using the Staph express disk. If you encounter high background flora in your staph testing, the 3M™ Petrifilm™ Staph Express Disk can be used to identify Staph aureus from all suspect colonies. The 3M Petrifilm Plate Advantage In today’s environment of heightened focus on food safety and increasingly stringent quality requirements, labs are under pressure to provide tests that are fast, reliable and consistently accurate. 3M Petrifilm Plates have demonstrated consistent performance that has been cited globally with more than 200 certificates, recognitions, validations, and peer-review publications. For more than 30 years, food safety professionals around the world have put their trust in 3M Petrifilm Plates. That’s because 3M Petrifilm Plates have streamlined, standardized and simplified the process of microbial quantitative indicator testing, improving productivity and helping ensure the highest levels of product quality. -

 Yeast & Mould results in 3-5 days This 3M Petrifilm Yeast and Mold Count Plate is a sample-ready-culture medium system which contains nutrients supplmented with antibiotics, a cold-water-soluble gelling agent, and an indicator that facilitates yeast and mold enumeration. Yeasts are easily differentiated from molds on the plate:
Yeast & Mould results in 3-5 days This 3M Petrifilm Yeast and Mold Count Plate is a sample-ready-culture medium system which contains nutrients supplmented with antibiotics, a cold-water-soluble gelling agent, and an indicator that facilitates yeast and mold enumeration. Yeasts are easily differentiated from molds on the plate:- Yeasts are typically indicated by small, blue-green colonies with defined edges and no foci.
- Molds are indicated by large, variably-colored colonies with diffuse edges and center foci.
-

 On-going Performance Monitoring for Routine Patient Testing AccuPlex Monkeypox Positive Reference Material Kit is a full-process quality solution designed to challenge the molecular test procedure from extraction to detection, ensuring clinical laboratories can have confidence in their assay results. The product contains positive materials directed against targets for Monkeypox assays.
On-going Performance Monitoring for Routine Patient Testing AccuPlex Monkeypox Positive Reference Material Kit is a full-process quality solution designed to challenge the molecular test procedure from extraction to detection, ensuring clinical laboratories can have confidence in their assay results. The product contains positive materials directed against targets for Monkeypox assays.- Non-infectious and replication deficient, enables safe handling in contrast to viral samples
- Fully-extractable with a real viral protein coat; superior to “naked” transcribed RNA
- Long shelf life: 2-year stability at 2 - 8°C
-

 SeraCare’s SARS-CoV-2 Quality Solution AccuPlex™ SARS-CoV-2 Reference Material, from SeraCare, is designed to measure day-to-day performance of the assay, providing both a positive and a negative reference solution. The product is available in versions that offer different SARS-CoV-2 sequence coverage: Version 1 covers the CDC and WHO consensus sequences, Version 2 has been enhanced to also include the S gene region, and Version 3 is expanded to include the full SARS‐CoV‐2 viral genome (Table 1). All products also include negative reference materials targeting the human RNase P gene. AccuPlex™ SARS-CoV-2 Verification Panel, coupled with the AccuPlex™ SARS-CoV-2 Reference Material, is a complete Quality Solution. Both products are available in three versions with different SARS-CoV-2 sequence coverage: Version 1 covers the CDC and WHO consensus sequences, Version 2 has been enhanced to also include the S gene region, and Version 3 is expanded to include the full SARS-CoV-2 viral genome (Table 1). All products also include negative reference materials targeting the human RNase P gene.
SeraCare’s SARS-CoV-2 Quality Solution AccuPlex™ SARS-CoV-2 Reference Material, from SeraCare, is designed to measure day-to-day performance of the assay, providing both a positive and a negative reference solution. The product is available in versions that offer different SARS-CoV-2 sequence coverage: Version 1 covers the CDC and WHO consensus sequences, Version 2 has been enhanced to also include the S gene region, and Version 3 is expanded to include the full SARS‐CoV‐2 viral genome (Table 1). All products also include negative reference materials targeting the human RNase P gene. AccuPlex™ SARS-CoV-2 Verification Panel, coupled with the AccuPlex™ SARS-CoV-2 Reference Material, is a complete Quality Solution. Both products are available in three versions with different SARS-CoV-2 sequence coverage: Version 1 covers the CDC and WHO consensus sequences, Version 2 has been enhanced to also include the S gene region, and Version 3 is expanded to include the full SARS-CoV-2 viral genome (Table 1). All products also include negative reference materials targeting the human RNase P gene.- Non-infectious and replication deficient, enables safe handling of positive material
- Fully-extractable with a real viral protein coat; serves as a full-process reference material
- Compatible with assays targeting CDC and WHO consensus sequences
- Includes negative reference material for targeting sequences for the human RNAse P gene
- 2 year stability at 2 - 8°C
- Customisable to sequences of interest to meet unique assay design requirements
-

 Clinical Diagnostic Quality Solutions for SARS-CoV-2 Variant Analysis AccuPlex SARS-CoV-2 Variant Panel 1 contains the full SARS-CoV-2 genomic RNA with a focus on S-gene mutations (Table 1) in three prominent variants of concern (VOC): UK variant B.1.1.7, South Africa variant B.1.351, and Brazil variant P.1. The kit has one vial for each variant as well as a fourth wild type (reference sequence NC_045512) control vial. An RNase P negative control vial is also included.
Clinical Diagnostic Quality Solutions for SARS-CoV-2 Variant Analysis AccuPlex SARS-CoV-2 Variant Panel 1 contains the full SARS-CoV-2 genomic RNA with a focus on S-gene mutations (Table 1) in three prominent variants of concern (VOC): UK variant B.1.1.7, South Africa variant B.1.351, and Brazil variant P.1. The kit has one vial for each variant as well as a fourth wild type (reference sequence NC_045512) control vial. An RNase P negative control vial is also included.- Performance confirmation of existing SARS-CoV-2 molecular assays with different variants
- Reference materials for SARS-CoV-2 variant genotyping assays
- Reference materials for SARS-CoV-2 NGS assays
- Non-infectious and replication deficient, enables safe handling of positive material
- Fully extractable with a real viral protein coat; superior to “naked” transcribed RNA
- 2 year stability at 2 - 8°C
-

 Full Genome Coverage now available AccuPlex™ SARS-CoV-2 Verification Panel, from SeraCare, is optimised for assay verification at installation by documenting test performance along the assay’s range enabling laboratories to establish lower limits of detection, perform assay comparisons, and evaluate staff proficiency. The product contains positive materials including the full SARS-CoV-2 viral genome, and negative materials targeting the human RNase P gene. AccuPlex™ SARS-CoV-2 Verification Panel, coupled with the AccuPlex™ SARS-CoV-2 Reference Material, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their SARS-CoV-2 assay results.
Full Genome Coverage now available AccuPlex™ SARS-CoV-2 Verification Panel, from SeraCare, is optimised for assay verification at installation by documenting test performance along the assay’s range enabling laboratories to establish lower limits of detection, perform assay comparisons, and evaluate staff proficiency. The product contains positive materials including the full SARS-CoV-2 viral genome, and negative materials targeting the human RNase P gene. AccuPlex™ SARS-CoV-2 Verification Panel, coupled with the AccuPlex™ SARS-CoV-2 Reference Material, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their SARS-CoV-2 assay results.- Non-infectious and replication deficient, enables safe handling of positive material
- Fully-extractable with a real viral protein coat; serves as a full-process reference material
- Compatible with assays targeting CDC and WHO consensus sequences
- Includes negative reference material for targeting sequences for the human RNAse P gene
- 2 year stability at 2 - 8°C
- Customisable to sequences of interest to meet unique assay design requirements
-

 Measure day-to-day performance AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, from SeraCare, is designed to measure day-to-day performance of the assay, providing both a positive and a negative reference solution. Designed for use with multiplexed molecular assays that can detect SARS-CoV-2, influenza A/B and respiratory syncytial virus (RSV). AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, coupled with the AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their multiplex assay results.
Measure day-to-day performance AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, from SeraCare, is designed to measure day-to-day performance of the assay, providing both a positive and a negative reference solution. Designed for use with multiplexed molecular assays that can detect SARS-CoV-2, influenza A/B and respiratory syncytial virus (RSV). AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, coupled with the AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their multiplex assay results.- Non-infectious and replication deficient, enables safe handling of positive material
- Fully-extractable with a real viral protein coat; serves as a full-process reference material
- 2 year stability at 2 - 8°C
- Optimised for assay verification and day-to-day performance monitoring
-

 Have confidence in your multiplex assay results for SARS-CoV-2 & Flu A/B AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, from SeraCare, is optimised for assay verification at installation by documenting test performance along the assay’s range, enabling laboratories to establish lower limits of detection, perform assay comparisons, and evaluate staff proficiency. Designed for use with multiplexed molecular assays that can detect SARS-CoV-2, influenza A/B and respiratory syncytial virus (RSV). AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, coupled with the AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their multiplex assay results.
Have confidence in your multiplex assay results for SARS-CoV-2 & Flu A/B AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, from SeraCare, is optimised for assay verification at installation by documenting test performance along the assay’s range, enabling laboratories to establish lower limits of detection, perform assay comparisons, and evaluate staff proficiency. Designed for use with multiplexed molecular assays that can detect SARS-CoV-2, influenza A/B and respiratory syncytial virus (RSV). AccuPlex™ SARS-CoV-2, Flu A/B and RSV Verification Panel, coupled with the AccuPlex™ SARS-CoV-2, Flu A/B and RSV Reference Material Kit, is a complete Quality Solution designed to challenge the entire molecular test procedure ensuring clinical laboratories can have confidence in their multiplex assay results.- Non-infectious and replication deficient, enables safe handling of positive material
- Fully-extractable with a real viral protein coat; serves as a full-process reference material
- 2 year stability at 2 - 8°C
- Optimised for assay verification and day-to-day performance monitoring
-

 Ensure antibody testing accuracy and performance To support the many SARS-CoV-2 serology assay formats available, the kit is currently offered in two configurations, one designed for IgG-specific assays and the other for total antibody tests. Series 1000 is released using the Abbott ARCHITECT SARS-CoV-2 IgG assay, and Series 2000 is released using the Roche cobas® Elecsys® Anti-SARS-CoV-2 assay. Reference materials are specifically formulated to exhibit low positive reactivity in true patient-like matrices, to test assay performance near critical signal cutoff boundaries. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterised human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting.
Ensure antibody testing accuracy and performance To support the many SARS-CoV-2 serology assay formats available, the kit is currently offered in two configurations, one designed for IgG-specific assays and the other for total antibody tests. Series 1000 is released using the Abbott ARCHITECT SARS-CoV-2 IgG assay, and Series 2000 is released using the Roche cobas® Elecsys® Anti-SARS-CoV-2 assay. Reference materials are specifically formulated to exhibit low positive reactivity in true patient-like matrices, to test assay performance near critical signal cutoff boundaries. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterised human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. -

 Ensure Strep A Laboratory Testing Assay Performance ACCURUN® reference materials are intended to estimate laboratory testing precision and can be used to detect errors in laboratory testing procedures. ACCURUN Swab Strep A Reference Material is formulated for use with molecular test methods used to detect Streptococcus pyogenes (Strep A). They are designed as whole organism reference materials that can be used to assess accuracy and performance of the full testing process – extraction, amplification, and detection – of molecular assays for Strep A. For Research Use Only. Not for use in diagnostic procedures.
Ensure Strep A Laboratory Testing Assay Performance ACCURUN® reference materials are intended to estimate laboratory testing precision and can be used to detect errors in laboratory testing procedures. ACCURUN Swab Strep A Reference Material is formulated for use with molecular test methods used to detect Streptococcus pyogenes (Strep A). They are designed as whole organism reference materials that can be used to assess accuracy and performance of the full testing process – extraction, amplification, and detection – of molecular assays for Strep A. For Research Use Only. Not for use in diagnostic procedures. -

 Whole-cell or whole-organism external controls Empower your results and challenge your methods with SeraCare’s ACCURUN® molecular controls and reference materials. ACCURUN molecular controls and reference materials are whole-cell or whole-organism external controls that help you monitor all aspects of your molecular testing methods and provide additional confidence in your laboratory test results. A well-designed QC program can help you avoid costly false-negative or false-positive results. ACCURUN molecular controls effectively detect low-positives closer to assay-specific cutoffs, enabling better detection of assay variability. Powerful ACCURUN controls and reference materials help to ensure complete control over assay performance monitoring. Monitors the Entire Testing Process. In order to execute the highest level of quality control, QC methods should be able to monitor the entire testing process, not just a portion. ACCURUN molecular controls are whole-cell or whole-organism controls that can appropriately challenge your assay from extraction through detection.
Whole-cell or whole-organism external controls Empower your results and challenge your methods with SeraCare’s ACCURUN® molecular controls and reference materials. ACCURUN molecular controls and reference materials are whole-cell or whole-organism external controls that help you monitor all aspects of your molecular testing methods and provide additional confidence in your laboratory test results. A well-designed QC program can help you avoid costly false-negative or false-positive results. ACCURUN molecular controls effectively detect low-positives closer to assay-specific cutoffs, enabling better detection of assay variability. Powerful ACCURUN controls and reference materials help to ensure complete control over assay performance monitoring. Monitors the Entire Testing Process. In order to execute the highest level of quality control, QC methods should be able to monitor the entire testing process, not just a portion. ACCURUN molecular controls are whole-cell or whole-organism controls that can appropriately challenge your assay from extraction through detection. -

 Empower your results and challenge your methods with SeraCare’s ACCURUN® serology controls and reference materials. SeraCare's ACCURUN controls and reference materials are designed to be weakly reactive to help monitor your serology assays and provide additional confidence in your laboratory test results. Monitoring your assay performance can help you avoid costly repeats and, more importantly, avoid false-negative and false-positive results. With ACCURUN controls, you can troubleshoot your test methods and isolate system errors in your laboratory. Powerful ACCURUN controls and reference materials help to ensure complete control over assay performance monitoring. Challenges Your Assay More Effectively A low-positive control challenges your assay more than a medium-to-high control since it is closer to the assay cut-off; therefore, any variability in the assay will be easier to detect. Assay monitoring with ACCURUN single and multi-analyte controls improves confidence in your serology test results.
Empower your results and challenge your methods with SeraCare’s ACCURUN® serology controls and reference materials. SeraCare's ACCURUN controls and reference materials are designed to be weakly reactive to help monitor your serology assays and provide additional confidence in your laboratory test results. Monitoring your assay performance can help you avoid costly repeats and, more importantly, avoid false-negative and false-positive results. With ACCURUN controls, you can troubleshoot your test methods and isolate system errors in your laboratory. Powerful ACCURUN controls and reference materials help to ensure complete control over assay performance monitoring. Challenges Your Assay More Effectively A low-positive control challenges your assay more than a medium-to-high control since it is closer to the assay cut-off; therefore, any variability in the assay will be easier to detect. Assay monitoring with ACCURUN single and multi-analyte controls improves confidence in your serology test results. -

 Available for use with serological and molecular assays, SeraCare’s portfolio of AccuSet performance panels contains highly characterized, raw, undiluted plasma specimens collected from unique individuals positive for your analytes of interest. Each panel contains a comprehensive comparative data sheet with test results from a wide variety of leading commercially available assays and platforms. AccuSet performance panels can be used to evaluate assay specificity, sensitivity, repeatability, and reproducibility to assist you in validating new test methods and equipment, run head-to-head assay comparisons, demonstrate lab proficiency, and train laboratory personnel.
Available for use with serological and molecular assays, SeraCare’s portfolio of AccuSet performance panels contains highly characterized, raw, undiluted plasma specimens collected from unique individuals positive for your analytes of interest. Each panel contains a comprehensive comparative data sheet with test results from a wide variety of leading commercially available assays and platforms. AccuSet performance panels can be used to evaluate assay specificity, sensitivity, repeatability, and reproducibility to assist you in validating new test methods and equipment, run head-to-head assay comparisons, demonstrate lab proficiency, and train laboratory personnel. -

 Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing.
Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing. -
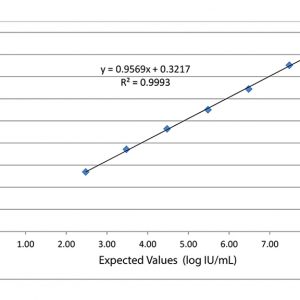
 Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration.
Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration. -

 Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program
Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program -

 Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms.
Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms. -

 Allergens in food can cause serious, life-threatening symptoms, even in small quantities in sensitive individuals. Thus, safe detection of even the smallest traces of an allergen in a final product is important for quality assurance in the food industry.There are 14 food ingredients responsible for 90% of all food allergies. Following EU Directive 2007/68/EC or 2000/13/EC, they must be reported on the package label when present in a product. In some countries limits exist, such as in Switzerland, where it is 1,000 ppm. For the detection of major food allergens, BIOTECON Diagnostics offers real-time PCR kits. When used in combination with the Allergen RM 800 reference material, quantification is also possible.
Allergens in food can cause serious, life-threatening symptoms, even in small quantities in sensitive individuals. Thus, safe detection of even the smallest traces of an allergen in a final product is important for quality assurance in the food industry.There are 14 food ingredients responsible for 90% of all food allergies. Following EU Directive 2007/68/EC or 2000/13/EC, they must be reported on the package label when present in a product. In some countries limits exist, such as in Switzerland, where it is 1,000 ppm. For the detection of major food allergens, BIOTECON Diagnostics offers real-time PCR kits. When used in combination with the Allergen RM 800 reference material, quantification is also possible. -

 Pre-reduced anaerobially sterilised (PRAS) plated and broth culture media Biolog Solutions for Anaerobic Culture Media Anaerobic organisms play critical roles in health and disease pathology of humans and animals, as well as throughout the Earth’s ecosystems. Isolating and culturing these fastidious organisms with consistent results requires specialised conditions. Your research can benefit from our decades of experience with formulating and manufacturing special nutritive TruPRAS™ media that never comes into contact with oxygen. We provide a wide range of media for specimen collection and transport, enriched plated agar media, and enriched liquid media specifically designed for culturing even organisms that were previously thought to be unculturable.
Pre-reduced anaerobially sterilised (PRAS) plated and broth culture media Biolog Solutions for Anaerobic Culture Media Anaerobic organisms play critical roles in health and disease pathology of humans and animals, as well as throughout the Earth’s ecosystems. Isolating and culturing these fastidious organisms with consistent results requires specialised conditions. Your research can benefit from our decades of experience with formulating and manufacturing special nutritive TruPRAS™ media that never comes into contact with oxygen. We provide a wide range of media for specimen collection and transport, enriched plated agar media, and enriched liquid media specifically designed for culturing even organisms that were previously thought to be unculturable. -

 The foodproof® Animal Identification Kits detect specific animal species in food by real-time PCR. They enable the identification and differentiation of swine, cattle, sheep, horse, donkey, chicken and goat in food, such as raw and heated meat, animal feed and animal products. Animal species detection in food protects product purity. It also controls against fraud through verification of the animal species used in meat products.
The foodproof® Animal Identification Kits detect specific animal species in food by real-time PCR. They enable the identification and differentiation of swine, cattle, sheep, horse, donkey, chicken and goat in food, such as raw and heated meat, animal feed and animal products. Animal species detection in food protects product purity. It also controls against fraud through verification of the animal species used in meat products. -

 Revolutionising Histopathology with Fully Automated Microtomy Automated microtomy is transforming the way histopathology laboratories operate—improving consistency, throughput, and diagnostic turnaround times. The Morphle Robotome (RTM30) represents a breakthrough in robotic microtomy and slide preparation, enabling laboratories to overcome traditional bottlenecks in tissue sectioning. Designed for modern cytology, histopathology, and cancer diagnostics laboratories, the Robotome automates the critical sectioning process—delivering high-quality, reproducible slides at scale, while reducing manual workload and variability. Key Applications: The Morphle Robotome is ideally suited for:
Revolutionising Histopathology with Fully Automated Microtomy Automated microtomy is transforming the way histopathology laboratories operate—improving consistency, throughput, and diagnostic turnaround times. The Morphle Robotome (RTM30) represents a breakthrough in robotic microtomy and slide preparation, enabling laboratories to overcome traditional bottlenecks in tissue sectioning. Designed for modern cytology, histopathology, and cancer diagnostics laboratories, the Robotome automates the critical sectioning process—delivering high-quality, reproducible slides at scale, while reducing manual workload and variability. Key Applications: The Morphle Robotome is ideally suited for:- Histopathology laboratories
- Cancer diagnostics centres
- Cytology workflows
- Biopharma and translational research labs
- High-volume diagnostic services
-

 Introducing the ILAS 4000 Robot The new ILAS 4000 robot is a unique solution to automate and standardise sample preparation for bacteria counters up to a maximum speed of 200 samples/hour. Fully refrigerated, samples are identified (RFID chips or barcode), homogenised, uncapped and analysed. The vials are automatically recapped after analysis and place back in their original tray position. Compatible with the BactoCount IBC Series: Bacteria & Somatic Cells Counter and alternative methods. The Standardisation Tray used for Laboratory Automation is compatible with the ILAS Robot.
Introducing the ILAS 4000 Robot The new ILAS 4000 robot is a unique solution to automate and standardise sample preparation for bacteria counters up to a maximum speed of 200 samples/hour. Fully refrigerated, samples are identified (RFID chips or barcode), homogenised, uncapped and analysed. The vials are automatically recapped after analysis and place back in their original tray position. Compatible with the BactoCount IBC Series: Bacteria & Somatic Cells Counter and alternative methods. The Standardisation Tray used for Laboratory Automation is compatible with the ILAS Robot. -

 Introducing the ILAS 3000 Robot The combination of the Bentley Instruments and high-tech robotics allows the complete automation and standardisation of the analytical chain and MSDs reduction. With the ILAS 3000 Robot, an operator can manage up to 3 lines simultaneously. Always with a goal of enhanced productivity and standardisation, this system analyses up to 600 samples/hour. Compatible with the Combi FTS Series: Infrared Analyser & Somatic Cells Counter and alternative methods. The Standardisation Tray used for Laboratory Automation is compatible with the ILAS Robot.
Introducing the ILAS 3000 Robot The combination of the Bentley Instruments and high-tech robotics allows the complete automation and standardisation of the analytical chain and MSDs reduction. With the ILAS 3000 Robot, an operator can manage up to 3 lines simultaneously. Always with a goal of enhanced productivity and standardisation, this system analyses up to 600 samples/hour. Compatible with the Combi FTS Series: Infrared Analyser & Somatic Cells Counter and alternative methods. The Standardisation Tray used for Laboratory Automation is compatible with the ILAS Robot. -

 Oral Car Kit Components Designed for use in Smaller Patients. The AVANOS* Mini Oral Care Advanced Kit is designed to provide comprehensive oral care for a 4 hour protocol. With small profile components, this kit is intended for use in patients with smaller mouths. Kits are complete with everything needed to provide comprehensive and consistent oral care, recognised as critical for preventing VAP by such leading healthcare organisations as the CDC, and AACN. Along with the BALLARD* Oral Care Mini Kit, AVANOS is addressing the known risk factors for VAP by offering a complete portfolio of Oral Care Kits, individual components and packs, designed by nurses, for nurses, to support both individual patient needs and hospital oral care protocols.
Oral Car Kit Components Designed for use in Smaller Patients. The AVANOS* Mini Oral Care Advanced Kit is designed to provide comprehensive oral care for a 4 hour protocol. With small profile components, this kit is intended for use in patients with smaller mouths. Kits are complete with everything needed to provide comprehensive and consistent oral care, recognised as critical for preventing VAP by such leading healthcare organisations as the CDC, and AACN. Along with the BALLARD* Oral Care Mini Kit, AVANOS is addressing the known risk factors for VAP by offering a complete portfolio of Oral Care Kits, individual components and packs, designed by nurses, for nurses, to support both individual patient needs and hospital oral care protocols.



