-

 Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -

 Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -
 Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Multichem D-Dimer Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is designed as a bi-level, liquid stable frozen product. The product will typically be run to provide a minimum of two levels of control to monitor D-Dimer assay performance within the analytical ranges. Multichem D-Dimer Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to -80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem D-Dimer Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is designed as a bi-level, liquid stable frozen product. The product will typically be run to provide a minimum of two levels of control to monitor D-Dimer assay performance within the analytical ranges. Multichem D-Dimer Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to -80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Third party, bi-level, liquid stable, multi-analyte quality control material to monitor the precision of laboratory testing procedures for Cerebral Spinal Fluid Assays. Multichem CSF, from Technopath Manufacturing Ltd, contains 4 analytes including CSF glucose, protein, IgG and lactate. Multichem CSF Controls are designed as bi-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls are should be stored at -20°C to -80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use.
Third party, bi-level, liquid stable, multi-analyte quality control material to monitor the precision of laboratory testing procedures for Cerebral Spinal Fluid Assays. Multichem CSF, from Technopath Manufacturing Ltd, contains 4 analytes including CSF glucose, protein, IgG and lactate. Multichem CSF Controls are designed as bi-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls are should be stored at -20°C to -80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use. -

 Multichem AMH Control, from Technopath Manufacturing Ltd, is intended for use as a third party, tri-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Anti Mullerian Hormone Assays. The product will typically be run to provide a minimum of 2 levels of control to monitor AMH assay performance within the analytical ranges. Multichem AMH Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem AMH Control, from Technopath Manufacturing Ltd, is intended for use as a third party, tri-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Anti Mullerian Hormone Assays. The product will typically be run to provide a minimum of 2 levels of control to monitor AMH assay performance within the analytical ranges. Multichem AMH Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem AE (Ammonia Ethanol) Control, from Technopath Manufacturing Ltd, is intended for use as a third party liquid stable quality control material to monitor the precision of laboratory testing procedures for Ammonia and Ethanol Assays. Multichem AE Control is designed as a liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem AE (Ammonia Ethanol) Control, from Technopath Manufacturing Ltd, is intended for use as a third party liquid stable quality control material to monitor the precision of laboratory testing procedures for Ammonia and Ethanol Assays. Multichem AE Control is designed as a liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem A1c Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Haemoglobin A1c determination Assays. The intended patient population are those with diabetes. The haemoglobin A1c test, also called HbA1c, glycated haemoglobin test, or glycol-haemoglobin, is an important blood test that shows how well a patient’s diabetes is being controlled. Multichem A1c Control is designed as a bi-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem A1c Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Haemoglobin A1c determination Assays. The intended patient population are those with diabetes. The haemoglobin A1c test, also called HbA1c, glycated haemoglobin test, or glycol-haemoglobin, is an important blood test that shows how well a patient’s diabetes is being controlled. Multichem A1c Control is designed as a bi-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Provides A Superior Tracheal Seal … Proven to Reduce Leakage AVANOS* MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are available in adult sizes with a Murphy Eye. They include a separate lumen with a dorsal opening above the cuff to provide access to the subglottic space. The subglottic space is reached via a normally open suction valve which includes a one-way port for rinsing the subglottic space with sterile saline (0.95% Sodium Chloride solution) or administering an air bolus to assist in maintaining suction lumen patency. Multiple size options available for the MICROCUFF* Subglottic Suctioning Adult Endotracheal Tube. Sizing options for the inner diameter vary between 7mm and 9mm. Similarly, the outer diameter sizing ranges from 10.4mm to 12.8mm. Cuff sizes come in 28mm and 30mm. The MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are manufactured from natural rubber that is latex-free. EO Sterilised, they should be used no more than once. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
Provides A Superior Tracheal Seal … Proven to Reduce Leakage AVANOS* MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are available in adult sizes with a Murphy Eye. They include a separate lumen with a dorsal opening above the cuff to provide access to the subglottic space. The subglottic space is reached via a normally open suction valve which includes a one-way port for rinsing the subglottic space with sterile saline (0.95% Sodium Chloride solution) or administering an air bolus to assist in maintaining suction lumen patency. Multiple size options available for the MICROCUFF* Subglottic Suctioning Adult Endotracheal Tube. Sizing options for the inner diameter vary between 7mm and 9mm. Similarly, the outer diameter sizing ranges from 10.4mm to 12.8mm. Cuff sizes come in 28mm and 30mm. The MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are manufactured from natural rubber that is latex-free. EO Sterilised, they should be used no more than once. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -

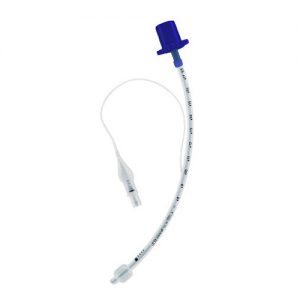 A new standard for paediatric airway management MICROCUFF* Paediatric Endotracheal Tubes from AVANOS, designed specifically for the paediatric anatomy, offer the advantages of a cuffed tube, reducing tracheal trauma and providing a sealed airway that allows minimal and low flow anaesthesia use.1 Its short and cylindrical cuff membrane compensates for different sized and shaped paediatric airways, reducing the need for replacing oversized tracheal tubes, and resulting in lower re-intubation rates.13 The MICROCUFF* brand has changed the existing green Vent Connector on all of the MICROCUFF* tubes and replaced it with a blue Vent Connector. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
A new standard for paediatric airway management MICROCUFF* Paediatric Endotracheal Tubes from AVANOS, designed specifically for the paediatric anatomy, offer the advantages of a cuffed tube, reducing tracheal trauma and providing a sealed airway that allows minimal and low flow anaesthesia use.1 Its short and cylindrical cuff membrane compensates for different sized and shaped paediatric airways, reducing the need for replacing oversized tracheal tubes, and resulting in lower re-intubation rates.13 The MICROCUFF* brand has changed the existing green Vent Connector on all of the MICROCUFF* tubes and replaced it with a blue Vent Connector. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -

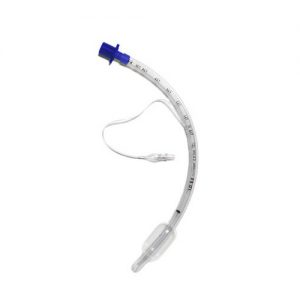 Provides A Superior Tracheal Seal … A Better Fit and Better Seal The MICROCUFF* tube from AVANOS, has advanced microthin polyurethane cuff material that allows the channels to “self-seal,” reducing the possibility of leakage. The build of the tube provides an effective seal at low cuff pressure. In turn, the MICROCUFF* may reduce micro-aspiration of potentially infectious pharyngeal secretions14. What’s more, it is designed for better contact with tracheal contour14. The thinner material enables greater visualisation of vocal cords when cuff is deflated. The MICROCUFF* is non-sterile and made from natural rubber that is latex-free. Intended for use in adults and for single-use. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
Provides A Superior Tracheal Seal … A Better Fit and Better Seal The MICROCUFF* tube from AVANOS, has advanced microthin polyurethane cuff material that allows the channels to “self-seal,” reducing the possibility of leakage. The build of the tube provides an effective seal at low cuff pressure. In turn, the MICROCUFF* may reduce micro-aspiration of potentially infectious pharyngeal secretions14. What’s more, it is designed for better contact with tracheal contour14. The thinner material enables greater visualisation of vocal cords when cuff is deflated. The MICROCUFF* is non-sterile and made from natural rubber that is latex-free. Intended for use in adults and for single-use. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -

 Solid Phase Cytometry meets Artificial Intelligence TECHNOPATH partners with Microbs for their fully automated solution. To boost productivity in your microbial QC lab, Microbs launches IAN®, an Ultra Rapid Microbiological Method allowing an accurate enumeration of bacteria, yeast and mould within minutes, not days! With IAN®, you can redesign your quality control. No need to wait days to check the microbiological quality of your production. IAN® gives you a quantitative measurement of your contamination in 15 minutes using our innovative direct non-growth detection method.
Solid Phase Cytometry meets Artificial Intelligence TECHNOPATH partners with Microbs for their fully automated solution. To boost productivity in your microbial QC lab, Microbs launches IAN®, an Ultra Rapid Microbiological Method allowing an accurate enumeration of bacteria, yeast and mould within minutes, not days! With IAN®, you can redesign your quality control. No need to wait days to check the microbiological quality of your production. IAN® gives you a quantitative measurement of your contamination in 15 minutes using our innovative direct non-growth detection method.- Mobile Laboratory - Autonomous, its fits where you need it.
- Speed - Time to result: 15 minutes, no enrichment needed.
- Simplicity - No expertise required
- Accuracy - Detection range from 1 to ≈ 5 000 microorganisms/volume analysed
- Liquid sample - Sampling from 100µL to 200mL
- Traceability - Full Data integrity, 21 CFR 11 compliant



