-

 Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia.
Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia. -

 Used for mild prolapse and stress incontinence The Silicone Ring with Knob Pessary is used for stress urinary incontinence with or without mild prolapse.
Used for mild prolapse and stress incontinence The Silicone Ring with Knob Pessary is used for stress urinary incontinence with or without mild prolapse. -

 Positive quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19 G is an unasssayed third-party quality control for Antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19 G (unassayed) is a positive control for antibodies to the SARS-CoV-2 virus (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®.
Positive quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19 G is an unasssayed third-party quality control for Antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19 G (unassayed) is a positive control for antibodies to the SARS-CoV-2 virus (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®. -

 Negative quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19Neg is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19Neg (unassayed) is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®.
Negative quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19Neg is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19Neg (unassayed) is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®. -

 Negative multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-SeroNeg provides a negative control for the analytes listed below. Scroll down to view the product benefits and analyte list. Multichem ID SeroNeg provides a third party, unassayed, negative control for commonly tested assays for anti-HIV, anti-HCV, anti HTLV, anti-HBc, anti-Treponema, HBsAg and HIVp24
Negative multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-SeroNeg provides a negative control for the analytes listed below. Scroll down to view the product benefits and analyte list. Multichem ID SeroNeg provides a third party, unassayed, negative control for commonly tested assays for anti-HIV, anti-HCV, anti HTLV, anti-HBc, anti-Treponema, HBsAg and HIVp24 -

 Positive multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-B is an unassayed control, optimised for Abbott and Diasorin instruments. Scroll down to view the product benefits and analyte list. The Multichem ID Multi-marker range of products is easily identifiable by their colour coding (in this case B for blue). Multichem ID-B consolidates six analytes (see analyte list below). This unassayed product is optimised for specific platforms, including; Abbott Architect, Abbott Alinity i, Abbott Alinity s, DiaSorin Liaison, DiaSorin Liaison XL MUREX.
Positive multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-B is an unassayed control, optimised for Abbott and Diasorin instruments. Scroll down to view the product benefits and analyte list. The Multichem ID Multi-marker range of products is easily identifiable by their colour coding (in this case B for blue). Multichem ID-B consolidates six analytes (see analyte list below). This unassayed product is optimised for specific platforms, including; Abbott Architect, Abbott Alinity i, Abbott Alinity s, DiaSorin Liaison, DiaSorin Liaison XL MUREX. -

 CURAPAX® CARE CONCEPT Single-Use Heating and Cooling Pads Hygienic, safe and timesaving heat and cold therapy Provide convenient heat and cold therapy for every hospital ward and make treatment easier, safer and more comfortable for both patients and caregivers. To be heated in microwave or incubator or to be cooled in a refrigerator
CURAPAX® CARE CONCEPT Single-Use Heating and Cooling Pads Hygienic, safe and timesaving heat and cold therapy Provide convenient heat and cold therapy for every hospital ward and make treatment easier, safer and more comfortable for both patients and caregivers. To be heated in microwave or incubator or to be cooled in a refrigerator- Hygienic single-use concept which reduces cross contamination
- Timesaving preparation and disposal
- A safe solution for both patients and staff
-

 Clinical Diagnostic Quality Solutions for SARS-CoV-2 Variant Analysis AccuPlex SARS-CoV-2 Variant Panel 1 contains the full SARS-CoV-2 genomic RNA with a focus on S-gene mutations (Table 1) in three prominent variants of concern (VOC): UK variant B.1.1.7, South Africa variant B.1.351, and Brazil variant P.1. The kit has one vial for each variant as well as a fourth wild type (reference sequence NC_045512) control vial. An RNase P negative control vial is also included.
Clinical Diagnostic Quality Solutions for SARS-CoV-2 Variant Analysis AccuPlex SARS-CoV-2 Variant Panel 1 contains the full SARS-CoV-2 genomic RNA with a focus on S-gene mutations (Table 1) in three prominent variants of concern (VOC): UK variant B.1.1.7, South Africa variant B.1.351, and Brazil variant P.1. The kit has one vial for each variant as well as a fourth wild type (reference sequence NC_045512) control vial. An RNase P negative control vial is also included.- Performance confirmation of existing SARS-CoV-2 molecular assays with different variants
- Reference materials for SARS-CoV-2 variant genotyping assays
- Reference materials for SARS-CoV-2 NGS assays
- Non-infectious and replication deficient, enables safe handling of positive material
- Fully extractable with a real viral protein coat; superior to “naked” transcribed RNA
- 2 year stability at 2 - 8°C
-

 Ensure antibody testing accuracy and performance To support the many SARS-CoV-2 serology assay formats available, the kit is currently offered in two configurations, one designed for IgG-specific assays and the other for total antibody tests. Series 1000 is released using the Abbott ARCHITECT SARS-CoV-2 IgG assay, and Series 2000 is released using the Roche cobas® Elecsys® Anti-SARS-CoV-2 assay. Reference materials are specifically formulated to exhibit low positive reactivity in true patient-like matrices, to test assay performance near critical signal cutoff boundaries. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterised human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting.
Ensure antibody testing accuracy and performance To support the many SARS-CoV-2 serology assay formats available, the kit is currently offered in two configurations, one designed for IgG-specific assays and the other for total antibody tests. Series 1000 is released using the Abbott ARCHITECT SARS-CoV-2 IgG assay, and Series 2000 is released using the Roche cobas® Elecsys® Anti-SARS-CoV-2 assay. Reference materials are specifically formulated to exhibit low positive reactivity in true patient-like matrices, to test assay performance near critical signal cutoff boundaries. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterised human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. -

 Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing.
Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing. -

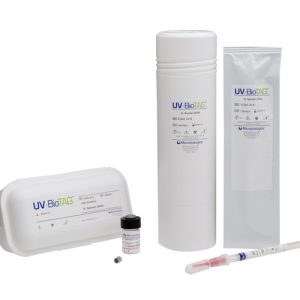 QC microorganisms with Green Fluorescent Protein (GFP) markers It’s not uncommon for food samples to inadvertently become contaminated with control strains used in the laboratory. This leads to false-positive results which can have severe financial and regulatory implications. UV-BioTAG, our line of reference strains containing green fluorescent protein (GFP) markers, make it easy to distinguish standard microorganism strains from laboratory control strains. Optimised for highly visible fluorescence, UV-BioTAG strains provide better stability because the GFP marker is integrated into the chromosome rather than the plasmid. With UV-BioTAG, you can quickly and reliably establish whether a positive test result can be traced back to a control strain cross-contamination issue, or if it is from another source. Applications & Test Methods
QC microorganisms with Green Fluorescent Protein (GFP) markers It’s not uncommon for food samples to inadvertently become contaminated with control strains used in the laboratory. This leads to false-positive results which can have severe financial and regulatory implications. UV-BioTAG, our line of reference strains containing green fluorescent protein (GFP) markers, make it easy to distinguish standard microorganism strains from laboratory control strains. Optimised for highly visible fluorescence, UV-BioTAG strains provide better stability because the GFP marker is integrated into the chromosome rather than the plasmid. With UV-BioTAG, you can quickly and reliably establish whether a positive test result can be traced back to a control strain cross-contamination issue, or if it is from another source. Applications & Test Methods- Enumeration Methods
- Detection Methods
- Demonstration of Capability (DOC)
- Media QC
- Validation/Verification
-

 Qualitative control strains for QC testing It doesn’t get much easier than the LYFO-DISK from Microbiologics®. Simply rehydrate LYFO DISK qualitative microorganism pellets with a diluent, such as phosphate buffer or saline, and you’re ready to inoculate your culture media. LYFO DISK, packaged in a glass vial containing 6 pellets of a single strain, offers a ton of flexibility. It's an ideal solution for many quality control applications including presence/absence testing, microbial identification methods, antimicrobial susceptibility testing, media QC, water testing and more! Applications & Test Methods
Qualitative control strains for QC testing It doesn’t get much easier than the LYFO-DISK from Microbiologics®. Simply rehydrate LYFO DISK qualitative microorganism pellets with a diluent, such as phosphate buffer or saline, and you’re ready to inoculate your culture media. LYFO DISK, packaged in a glass vial containing 6 pellets of a single strain, offers a ton of flexibility. It's an ideal solution for many quality control applications including presence/absence testing, microbial identification methods, antimicrobial susceptibility testing, media QC, water testing and more! Applications & Test Methods- Culture Purposes
- Media and reagent QC
- QC of Antimicrobial Susceptibility Test
- QC of microbial identification including biochemical, PCR and rapid molecular methods
- Water tests (e.g. enzyme substrates)
- Disinfectant studies
- Verifications and Validations
- Proficiency tests
- Daily QC



