- Staphylococcus aureus subsp. aureus derived from ATCC® 6538™*
- Pseudomonas aeruginosa derived from ATCC® 9027™*
- Bacillus subtilis subsp. spizizenii derived from ATCC® 6633™*
- Candida albicans derived from ATCC® 10231™*
- Aspergillus brasiliensis derived from ATCC® 16404™*
- Escherichia coli derived from ATCC® 8739™*
-

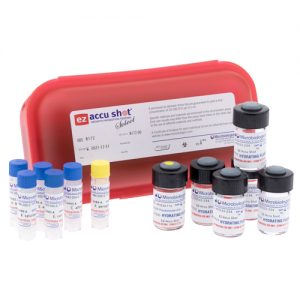 Five compendial GPT strains, plus E. coli – all in one convenient kit EZ-Accu Shot Select, from Microbiologics®, contains the 5 compendial organisms for Growth Promotion Testing, plus E. coli for a total of 6 strains in one convenient kit. Perfect for qualifying EZ-Accu Shot for use in your laboratory, the EZ-Accu Shot Select kit contains one instant dissolve pellet of each of the 6 strains and 6 vials of hydration fluid. Each pellet delivers 10-100 CFU per inoculum with no serial dilutions and provided 8 hours of stability after rehydration for added flexibility to perform tests. EZ-Accu Shot Select strains are just 3 passages from the reference culture to meet Pharmacopeia standards. Strains Include:
Five compendial GPT strains, plus E. coli – all in one convenient kit EZ-Accu Shot Select, from Microbiologics®, contains the 5 compendial organisms for Growth Promotion Testing, plus E. coli for a total of 6 strains in one convenient kit. Perfect for qualifying EZ-Accu Shot for use in your laboratory, the EZ-Accu Shot Select kit contains one instant dissolve pellet of each of the 6 strains and 6 vials of hydration fluid. Each pellet delivers 10-100 CFU per inoculum with no serial dilutions and provided 8 hours of stability after rehydration for added flexibility to perform tests. EZ-Accu Shot Select strains are just 3 passages from the reference culture to meet Pharmacopeia standards. Strains Include: -

 Economical Quantitative QC Microorganisms for Growth Promotion Testing EZ-CFU, from Microbiologics®, ready-to-use reference cultures for Growth Promotion Testing. Delivering 10-100 CFU per 0.1 ml inoculum, following a simple 1:10 dilution step, each microorganism suspension provides up to 90 tests. These handy kits allow for fast, reliable Growth Promotion Testing. Plus, EZ-CFU microorganisms are just three passages from the reference strains. All in all, EZ-CFU makes meeting Pharmacopeia standards for Growth Promotion Testing easier than ever. Applications & Test Methods
Economical Quantitative QC Microorganisms for Growth Promotion Testing EZ-CFU, from Microbiologics®, ready-to-use reference cultures for Growth Promotion Testing. Delivering 10-100 CFU per 0.1 ml inoculum, following a simple 1:10 dilution step, each microorganism suspension provides up to 90 tests. These handy kits allow for fast, reliable Growth Promotion Testing. Plus, EZ-CFU microorganisms are just three passages from the reference strains. All in all, EZ-CFU makes meeting Pharmacopeia standards for Growth Promotion Testing easier than ever. Applications & Test Methods- Growth Promotion Testing
- Media Challenge Testing
- Suitability of Counting Methods
- Suitability of Sterility Tests
- Suitability of Tests for Specified Microorganisms
- Microbial Limits Testing
- Microbial Enumeration Testing
- Validation of Neutralisation Methods
- Methods requiring a low CFU concentration
-

 Ready-to-Use Quantitative QC Microorganisms for Growth Promotion Testing Introducing EZ-CFU One Step, from Microbiologics®. This product is designed specifically for Growth Promotion Testing, Suitability of Tests for Specified Microorganisms, Suitability of Counting Methods and more. These ready-to-use quantitative microorganisms are just 3 passages from the reference culture and deliver 10-100 CFU per inoculum, as required by the Pharmacopeias, with minimal prep time. Applications & Test Methods
Ready-to-Use Quantitative QC Microorganisms for Growth Promotion Testing Introducing EZ-CFU One Step, from Microbiologics®. This product is designed specifically for Growth Promotion Testing, Suitability of Tests for Specified Microorganisms, Suitability of Counting Methods and more. These ready-to-use quantitative microorganisms are just 3 passages from the reference culture and deliver 10-100 CFU per inoculum, as required by the Pharmacopeias, with minimal prep time. Applications & Test Methods- Growth Promotion Testing
- Media Challenge Testing
- Suitability of Counting Methods
- Suitability of Sterility Tests
- Suitability of Tests for Specified Microorganisms
- Microbial Limits Testing
- Microbial Enumeration Testing
- Validation of Neutralisation Methods
- Methods requiring a low CFU concentration
-

 Ready-to-Use Microorganisms for Antimicrobial Effectiveness and Preservative Efficacy Challenge Testing Microbiologists are no strangers to the fact that safety and effectiveness are crucial elements of developing pharmaceutical and personal care products. The EZ-PEC™ quantitative microorganisms, from Microbiologics®, help laboratories conduct Antimicrobial Effectiveness and Preservative Efficacy testing with convenience and confidence. Following a few simple steps, EZ-PEC™ delivers a final concentration of 1.0 x 105 to 1.0 x 106 CFU per ml of the product being tested. It’s a simple solution that can save you time and resources while helping you meet Pharmacopeia and other standards and guidelines. Applications & Test Methods
Ready-to-Use Microorganisms for Antimicrobial Effectiveness and Preservative Efficacy Challenge Testing Microbiologists are no strangers to the fact that safety and effectiveness are crucial elements of developing pharmaceutical and personal care products. The EZ-PEC™ quantitative microorganisms, from Microbiologics®, help laboratories conduct Antimicrobial Effectiveness and Preservative Efficacy testing with convenience and confidence. Following a few simple steps, EZ-PEC™ delivers a final concentration of 1.0 x 105 to 1.0 x 106 CFU per ml of the product being tested. It’s a simple solution that can save you time and resources while helping you meet Pharmacopeia and other standards and guidelines. Applications & Test Methods- Antimicrobial Effectiveness Testing
- Preservative Efficacy Testing
- Applications requiring a high CFU concentration
-

 Quantitative spore-forming QC microorganisms for spoilage detection methods Introducing EZ-SPORE, from Microbiologics®. Spore-forming pathogens can present unique challenges for food laboratories that are conducting tests to detect these contaminants. That’s why food microbiology labs across the globe rely on EZ-SPORE quantitative microorganism preparations for validating their spoilage detection methods or for disinfectant qualification studies. EZ-SPORE preparations provide a concentration of 104 CFU per pellet, making microbiological challenge testing for spore-formers quicker and easier. Applications & Test Methods
Quantitative spore-forming QC microorganisms for spoilage detection methods Introducing EZ-SPORE, from Microbiologics®. Spore-forming pathogens can present unique challenges for food laboratories that are conducting tests to detect these contaminants. That’s why food microbiology labs across the globe rely on EZ-SPORE quantitative microorganism preparations for validating their spoilage detection methods or for disinfectant qualification studies. EZ-SPORE preparations provide a concentration of 104 CFU per pellet, making microbiological challenge testing for spore-formers quicker and easier. Applications & Test Methods- Daily food process controls
- Spoilage detection methods
- Disinfectant Qualification Studies
-

 Helping microbiologists spread culture since 1986 Introducing KWIK-STICK, from Microbiologics®. Laboratories throughout the world have been praising the KWIK-STIK for decades. That’s because its simple, all-inclusive design makes life easier for lab techs while reducing chances for errors. Each KWIK-STIK contains a qualitative lyophilised microorganism pellet, ampoule of hydrating fluid and inoculating swab. Everything you need to grow reference cultures for QC testing is included in this one handy device. Available in packs of 2 or 6, KWIK-STIKs are universally used for all types of microbiological control testing Applications & Test Methods
Helping microbiologists spread culture since 1986 Introducing KWIK-STICK, from Microbiologics®. Laboratories throughout the world have been praising the KWIK-STIK for decades. That’s because its simple, all-inclusive design makes life easier for lab techs while reducing chances for errors. Each KWIK-STIK contains a qualitative lyophilised microorganism pellet, ampoule of hydrating fluid and inoculating swab. Everything you need to grow reference cultures for QC testing is included in this one handy device. Available in packs of 2 or 6, KWIK-STIKs are universally used for all types of microbiological control testing Applications & Test Methods- Culture Purposes
Daily QC[/fusion_li_item]
- Verification and Validation
- QC of Identification Systems and Assay Kits
- QC of Antimicrobial Susceptibility Test
-

 Two passage reference cultures in our signature KWIK-STIK format Introducing KWIK-STICK Plus, from Microbiologics®. Some laboratory applications require strains that are a fewer passages from the original reference culture. For those special instances, we offer KWIK-STIK Plus, which combines microorganisms that are just two passages from their reference strains, with our signature KWIK-STIK format. Each device features a qualitative lyophilised microorganism pellet, ampoule of hydrating fluid and inoculating swab; so everything you need for growing control cultures is included in one handy device. Applications & Test Methods
Two passage reference cultures in our signature KWIK-STIK format Introducing KWIK-STICK Plus, from Microbiologics®. Some laboratory applications require strains that are a fewer passages from the original reference culture. For those special instances, we offer KWIK-STIK Plus, which combines microorganisms that are just two passages from their reference strains, with our signature KWIK-STIK format. Each device features a qualitative lyophilised microorganism pellet, ampoule of hydrating fluid and inoculating swab; so everything you need for growing control cultures is included in one handy device. Applications & Test Methods- Antibiotic assays
- Instrument validations
- Validation of neutralization methods
- Water tests
- Disinfectant qualification
-

 Qualitative control strains as Certified Reference Material for ISO 17025 accredited labs Increasing regulatory oversight in food, pharmaceutical and water industries has prompted many microbiology labs to seek accreditation to ISO 17025 standards. The Lab-Elite CRM, from Microbiologics®, is designed to help those labs meet the requirements of this standard which specify that “reference materials shall, where possible, be traceable to SI units of measurements, or to Certified Reference Materials (section 5.6.3.2)”. Lab-Elite CRM microorganisms are just one passage from the reference culture and are delivered in our signature KWIK-STIK format. As a Certified Reference Material, Lab-Elite comes with a comprehensive Certificate of Analysis detailing the strains identity and characteristics. Applications & Test Methods
Qualitative control strains as Certified Reference Material for ISO 17025 accredited labs Increasing regulatory oversight in food, pharmaceutical and water industries has prompted many microbiology labs to seek accreditation to ISO 17025 standards. The Lab-Elite CRM, from Microbiologics®, is designed to help those labs meet the requirements of this standard which specify that “reference materials shall, where possible, be traceable to SI units of measurements, or to Certified Reference Materials (section 5.6.3.2)”. Lab-Elite CRM microorganisms are just one passage from the reference culture and are delivered in our signature KWIK-STIK format. As a Certified Reference Material, Lab-Elite comes with a comprehensive Certificate of Analysis detailing the strains identity and characteristics. Applications & Test Methods- ISO 17025 laboratories requiring CRM
- Validation/verification of processes and instruments
- Presence/Absence
- Identification methods
-

 Qualitative control strains for QC testing It doesn’t get much easier than the LYFO-DISK from Microbiologics®. Simply rehydrate LYFO DISK qualitative microorganism pellets with a diluent, such as phosphate buffer or saline, and you’re ready to inoculate your culture media. LYFO DISK, packaged in a glass vial containing 6 pellets of a single strain, offers a ton of flexibility. It's an ideal solution for many quality control applications including presence/absence testing, microbial identification methods, antimicrobial susceptibility testing, media QC, water testing and more! Applications & Test Methods
Qualitative control strains for QC testing It doesn’t get much easier than the LYFO-DISK from Microbiologics®. Simply rehydrate LYFO DISK qualitative microorganism pellets with a diluent, such as phosphate buffer or saline, and you’re ready to inoculate your culture media. LYFO DISK, packaged in a glass vial containing 6 pellets of a single strain, offers a ton of flexibility. It's an ideal solution for many quality control applications including presence/absence testing, microbial identification methods, antimicrobial susceptibility testing, media QC, water testing and more! Applications & Test Methods- Culture Purposes
- Media and reagent QC
- QC of Antimicrobial Susceptibility Test
- QC of microbial identification including biochemical, PCR and rapid molecular methods
- Water tests (e.g. enzyme substrates)
- Disinfectant studies
- Verifications and Validations
- Proficiency tests
- Daily QC
-

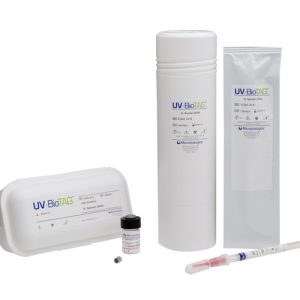 QC microorganisms with Green Fluorescent Protein (GFP) markers It’s not uncommon for food samples to inadvertently become contaminated with control strains used in the laboratory. This leads to false-positive results which can have severe financial and regulatory implications. UV-BioTAG, our line of reference strains containing green fluorescent protein (GFP) markers, make it easy to distinguish standard microorganism strains from laboratory control strains. Optimised for highly visible fluorescence, UV-BioTAG strains provide better stability because the GFP marker is integrated into the chromosome rather than the plasmid. With UV-BioTAG, you can quickly and reliably establish whether a positive test result can be traced back to a control strain cross-contamination issue, or if it is from another source. Applications & Test Methods
QC microorganisms with Green Fluorescent Protein (GFP) markers It’s not uncommon for food samples to inadvertently become contaminated with control strains used in the laboratory. This leads to false-positive results which can have severe financial and regulatory implications. UV-BioTAG, our line of reference strains containing green fluorescent protein (GFP) markers, make it easy to distinguish standard microorganism strains from laboratory control strains. Optimised for highly visible fluorescence, UV-BioTAG strains provide better stability because the GFP marker is integrated into the chromosome rather than the plasmid. With UV-BioTAG, you can quickly and reliably establish whether a positive test result can be traced back to a control strain cross-contamination issue, or if it is from another source. Applications & Test Methods- Enumeration Methods
- Detection Methods
- Demonstration of Capability (DOC)
- Media QC
- Validation/Verification
-

 PreBioM™ Microplates Introducing The PreBioM™ line of microplates from Biolog. The microplates allow researchers studying human or animal gut microbiome communities to better understand metabolism of prebiotics. PreBioM plates offer a multidimensional phenotype profiling solution based on the impact of prebiotics on microbial function. The three-plate system contains 90 unique prebiotic substrates tailored to investigate the intricate interplay between prebiotics and the microbiome. These plates consist of a curated selection of prebiotic substrates of various molecular weights designed to characterise bacterial species and their metabolic pathways. The PreBioM plates, combined with the Biolog Odin family of instruments and software, provide a high throughput phenotypic solution compatible with a wide range of species, including the obligate anaerobes inhabiting the gut microbiome as well as microbial communities.
PreBioM™ Microplates Introducing The PreBioM™ line of microplates from Biolog. The microplates allow researchers studying human or animal gut microbiome communities to better understand metabolism of prebiotics. PreBioM plates offer a multidimensional phenotype profiling solution based on the impact of prebiotics on microbial function. The three-plate system contains 90 unique prebiotic substrates tailored to investigate the intricate interplay between prebiotics and the microbiome. These plates consist of a curated selection of prebiotic substrates of various molecular weights designed to characterise bacterial species and their metabolic pathways. The PreBioM plates, combined with the Biolog Odin family of instruments and software, provide a high throughput phenotypic solution compatible with a wide range of species, including the obligate anaerobes inhabiting the gut microbiome as well as microbial communities. -

 Solid Phase Cytometry meets Artificial Intelligence TECHNOPATH partners with Microbs for their fully automated solution. To boost productivity in your microbial QC lab, Microbs launches IAN®, an Ultra Rapid Microbiological Method allowing an accurate enumeration of bacteria, yeast and mould within minutes, not days! With IAN®, you can redesign your quality control. No need to wait days to check the microbiological quality of your production. IAN® gives you a quantitative measurement of your contamination in 15 minutes using our innovative direct non-growth detection method.
Solid Phase Cytometry meets Artificial Intelligence TECHNOPATH partners with Microbs for their fully automated solution. To boost productivity in your microbial QC lab, Microbs launches IAN®, an Ultra Rapid Microbiological Method allowing an accurate enumeration of bacteria, yeast and mould within minutes, not days! With IAN®, you can redesign your quality control. No need to wait days to check the microbiological quality of your production. IAN® gives you a quantitative measurement of your contamination in 15 minutes using our innovative direct non-growth detection method.- Mobile Laboratory - Autonomous, its fits where you need it.
- Speed - Time to result: 15 minutes, no enrichment needed.
- Simplicity - No expertise required
- Accuracy - Detection range from 1 to ≈ 5 000 microorganisms/volume analysed
- Liquid sample - Sampling from 100µL to 200mL
- Traceability - Full Data integrity, 21 CFR 11 compliant
-

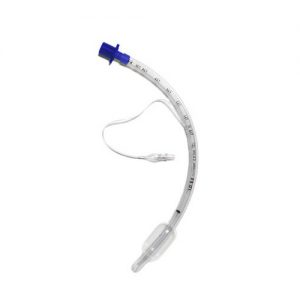 Provides A Superior Tracheal Seal … A Better Fit and Better Seal The MICROCUFF* tube from AVANOS, has advanced microthin polyurethane cuff material that allows the channels to “self-seal,” reducing the possibility of leakage. The build of the tube provides an effective seal at low cuff pressure. In turn, the MICROCUFF* may reduce micro-aspiration of potentially infectious pharyngeal secretions14. What’s more, it is designed for better contact with tracheal contour14. The thinner material enables greater visualisation of vocal cords when cuff is deflated. The MICROCUFF* is non-sterile and made from natural rubber that is latex-free. Intended for use in adults and for single-use. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
Provides A Superior Tracheal Seal … A Better Fit and Better Seal The MICROCUFF* tube from AVANOS, has advanced microthin polyurethane cuff material that allows the channels to “self-seal,” reducing the possibility of leakage. The build of the tube provides an effective seal at low cuff pressure. In turn, the MICROCUFF* may reduce micro-aspiration of potentially infectious pharyngeal secretions14. What’s more, it is designed for better contact with tracheal contour14. The thinner material enables greater visualisation of vocal cords when cuff is deflated. The MICROCUFF* is non-sterile and made from natural rubber that is latex-free. Intended for use in adults and for single-use. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -

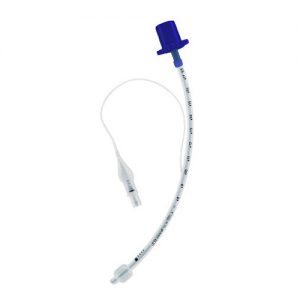 A new standard for paediatric airway management MICROCUFF* Paediatric Endotracheal Tubes from AVANOS, designed specifically for the paediatric anatomy, offer the advantages of a cuffed tube, reducing tracheal trauma and providing a sealed airway that allows minimal and low flow anaesthesia use.1 Its short and cylindrical cuff membrane compensates for different sized and shaped paediatric airways, reducing the need for replacing oversized tracheal tubes, and resulting in lower re-intubation rates.13 The MICROCUFF* brand has changed the existing green Vent Connector on all of the MICROCUFF* tubes and replaced it with a blue Vent Connector. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
A new standard for paediatric airway management MICROCUFF* Paediatric Endotracheal Tubes from AVANOS, designed specifically for the paediatric anatomy, offer the advantages of a cuffed tube, reducing tracheal trauma and providing a sealed airway that allows minimal and low flow anaesthesia use.1 Its short and cylindrical cuff membrane compensates for different sized and shaped paediatric airways, reducing the need for replacing oversized tracheal tubes, and resulting in lower re-intubation rates.13 The MICROCUFF* brand has changed the existing green Vent Connector on all of the MICROCUFF* tubes and replaced it with a blue Vent Connector. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -

 Provides A Superior Tracheal Seal … Proven to Reduce Leakage AVANOS* MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are available in adult sizes with a Murphy Eye. They include a separate lumen with a dorsal opening above the cuff to provide access to the subglottic space. The subglottic space is reached via a normally open suction valve which includes a one-way port for rinsing the subglottic space with sterile saline (0.95% Sodium Chloride solution) or administering an air bolus to assist in maintaining suction lumen patency. Multiple size options available for the MICROCUFF* Subglottic Suctioning Adult Endotracheal Tube. Sizing options for the inner diameter vary between 7mm and 9mm. Similarly, the outer diameter sizing ranges from 10.4mm to 12.8mm. Cuff sizes come in 28mm and 30mm. The MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are manufactured from natural rubber that is latex-free. EO Sterilised, they should be used no more than once. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here
Provides A Superior Tracheal Seal … Proven to Reduce Leakage AVANOS* MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are available in adult sizes with a Murphy Eye. They include a separate lumen with a dorsal opening above the cuff to provide access to the subglottic space. The subglottic space is reached via a normally open suction valve which includes a one-way port for rinsing the subglottic space with sterile saline (0.95% Sodium Chloride solution) or administering an air bolus to assist in maintaining suction lumen patency. Multiple size options available for the MICROCUFF* Subglottic Suctioning Adult Endotracheal Tube. Sizing options for the inner diameter vary between 7mm and 9mm. Similarly, the outer diameter sizing ranges from 10.4mm to 12.8mm. Cuff sizes come in 28mm and 30mm. The MICROCUFF* Subglottic Suctioning Adult Endotracheal Tubes are manufactured from natural rubber that is latex-free. EO Sterilised, they should be used no more than once. REFS: For references please refer to attached brochure - View MICROCUFF* Endotracheal Tubes Brochure here -
 Multichem A1c Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Haemoglobin A1c determination Assays. The intended patient population are those with diabetes. The haemoglobin A1c test, also called HbA1c, glycated haemoglobin test, or glycol-haemoglobin, is an important blood test that shows how well a patient’s diabetes is being controlled. Multichem A1c Control is designed as a bi-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem A1c Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Haemoglobin A1c determination Assays. The intended patient population are those with diabetes. The haemoglobin A1c test, also called HbA1c, glycated haemoglobin test, or glycol-haemoglobin, is an important blood test that shows how well a patient’s diabetes is being controlled. Multichem A1c Control is designed as a bi-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem AE (Ammonia Ethanol) Control, from Technopath Manufacturing Ltd, is intended for use as a third party liquid stable quality control material to monitor the precision of laboratory testing procedures for Ammonia and Ethanol Assays. Multichem AE Control is designed as a liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem AE (Ammonia Ethanol) Control, from Technopath Manufacturing Ltd, is intended for use as a third party liquid stable quality control material to monitor the precision of laboratory testing procedures for Ammonia and Ethanol Assays. Multichem AE Control is designed as a liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Multichem AMH Control, from Technopath Manufacturing Ltd, is intended for use as a third party, tri-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Anti Mullerian Hormone Assays. The product will typically be run to provide a minimum of 2 levels of control to monitor AMH assay performance within the analytical ranges. Multichem AMH Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem AMH Control, from Technopath Manufacturing Ltd, is intended for use as a third party, tri-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Anti Mullerian Hormone Assays. The product will typically be run to provide a minimum of 2 levels of control to monitor AMH assay performance within the analytical ranges. Multichem AMH Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Third party, bi-level, liquid stable, multi-analyte quality control material to monitor the precision of laboratory testing procedures for Cerebral Spinal Fluid Assays. Multichem CSF, from Technopath Manufacturing Ltd, contains 4 analytes including CSF glucose, protein, IgG and lactate. Multichem CSF Controls are designed as bi-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls are should be stored at -20°C to -80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use.
Third party, bi-level, liquid stable, multi-analyte quality control material to monitor the precision of laboratory testing procedures for Cerebral Spinal Fluid Assays. Multichem CSF, from Technopath Manufacturing Ltd, contains 4 analytes including CSF glucose, protein, IgG and lactate. Multichem CSF Controls are designed as bi-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls are should be stored at -20°C to -80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use. -

 Multichem D-Dimer Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is designed as a bi-level, liquid stable frozen product. The product will typically be run to provide a minimum of two levels of control to monitor D-Dimer assay performance within the analytical ranges. Multichem D-Dimer Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to -80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem D-Dimer Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is intended for use as a third party, bi-level, liquid stable quality control material to monitor the precision of laboratory testing procedures for D-Dimer Assays. Multichem D-Dimer Control is designed as a bi-level, liquid stable frozen product. The product will typically be run to provide a minimum of two levels of control to monitor D-Dimer assay performance within the analytical ranges. Multichem D-Dimer Control will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The Control is to be stored at -20°C to -80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -

 Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -
 Multichem IA Speciality Immunoassay Controls are intended for use as third party, tri- level, liquid stable, multi-analyte quality control materials to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Speciality, from Technopath Manufacturing Ltd, is designed to complement Multichem IA and Multichem IA Plus Immunoassay Controls by offering significantly increased stability for BNP, PTH and ACTH. In addition to these analytes, Multichem IA Speciality Controls also contains and provides tri level utility for Procalcitonin and Calcitonin. Multichem IA Speciality Immunoassay Controls are designed as tri-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls should be stored at -20°C to –80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use.
Multichem IA Speciality Immunoassay Controls are intended for use as third party, tri- level, liquid stable, multi-analyte quality control materials to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Speciality, from Technopath Manufacturing Ltd, is designed to complement Multichem IA and Multichem IA Plus Immunoassay Controls by offering significantly increased stability for BNP, PTH and ACTH. In addition to these analytes, Multichem IA Speciality Controls also contains and provides tri level utility for Procalcitonin and Calcitonin. Multichem IA Speciality Immunoassay Controls are designed as tri-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls should be stored at -20°C to –80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use. -

 Positive multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-B is an unassayed control, optimised for Abbott and Diasorin instruments. Scroll down to view the product benefits and analyte list. The Multichem ID Multi-marker range of products is easily identifiable by their colour coding (in this case B for blue). Multichem ID-B consolidates six analytes (see analyte list below). This unassayed product is optimised for specific platforms, including; Abbott Architect, Abbott Alinity i, Abbott Alinity s, DiaSorin Liaison, DiaSorin Liaison XL MUREX.
Positive multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-B is an unassayed control, optimised for Abbott and Diasorin instruments. Scroll down to view the product benefits and analyte list. The Multichem ID Multi-marker range of products is easily identifiable by their colour coding (in this case B for blue). Multichem ID-B consolidates six analytes (see analyte list below). This unassayed product is optimised for specific platforms, including; Abbott Architect, Abbott Alinity i, Abbott Alinity s, DiaSorin Liaison, DiaSorin Liaison XL MUREX. -

 Positive quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19 G is an unasssayed third-party quality control for Antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19 G (unassayed) is a positive control for antibodies to the SARS-CoV-2 virus (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®.
Positive quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19 G is an unasssayed third-party quality control for Antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19 G (unassayed) is a positive control for antibodies to the SARS-CoV-2 virus (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®. -

 Negative quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19Neg is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19Neg (unassayed) is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®.
Negative quality control for antibodies to SARS-CoV-2 (including IgG) Multichem ID-COVID19Neg is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). This Quality Control is available with access to the Technopath Clinical Diagnostics inter-laboratory Peer program, IAMQC®. The Multichem ID-COVID19Neg (unassayed) is an unassayed negative third-party quality control for antibodies to SARS-CoV-2 (including IgG). The kit is delivered in a choice of two configurations: 4x2mL tubes with an integrated instrument barcode and secondly 4 x 4mL vials. The product is suitable for use on all major platforms, including amongst others, Abbott, Roche and Siemens. All Multichem ID-COVID19 quality controls are supported by the inter-laboratory Peer program - IAMQC®. -

 Negative multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-SeroNeg provides a negative control for the analytes listed below. Scroll down to view the product benefits and analyte list. Multichem ID SeroNeg provides a third party, unassayed, negative control for commonly tested assays for anti-HIV, anti-HCV, anti HTLV, anti-HBc, anti-Treponema, HBsAg and HIVp24
Negative multi-marker QC Multichem Infectious Disease (ID) Quality Controls (QC) samples, have been specifically optimised and validated to match assays commonly used for infectious diseases testing. Multichem ID-SeroNeg provides a negative control for the analytes listed below. Scroll down to view the product benefits and analyte list. Multichem ID SeroNeg provides a third party, unassayed, negative control for commonly tested assays for anti-HIV, anti-HCV, anti HTLV, anti-HBc, anti-Treponema, HBsAg and HIVp24 -

 Third party, single elevated level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Bilirubin and Theophylline Assays. Multichem NB (Neonatal Bilirubin) Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Bilirubin and Theophylline Assays. An important patient population is Neonates, where a requirement has been identified for testing conditions associated with Neonatal hyperbilirubinemia. The product also contains Theophylline, a therapeutic drug that is given to Neonates to improve lung capacity and where it would be required to monitor for toxicity risk. Caffeine is a listed analyte that is added gravimetrically, and this has a clinical function as the primary treatment of the breathing disorders apnea of prematurity. Another important population is with any patient that has liver failure. It is important if the laboratory tests hyperbilirubinemia patient samples that they analyze QC regularly to monitor this range to ensure accuracy and precision at these elevated levels. The requirement for a high concentration of Bilirubin presents a conflict for consolidation of multi-analytes. This means that not only is a separate paediatric control required, but a control matrix that is treated with antioxidants to inhibit the oxidation of Bilirubin. The application of the most appropriate measures to reduce the effect of oxidation on the high levels of Bilirubin in the Control have been researched to ensure that open vial and closed stability is maintained. Multichem NB Control is designed as a single level, liquid stable frozen product. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Third party, single elevated level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Bilirubin and Theophylline Assays. Multichem NB (Neonatal Bilirubin) Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Bilirubin and Theophylline Assays. An important patient population is Neonates, where a requirement has been identified for testing conditions associated with Neonatal hyperbilirubinemia. The product also contains Theophylline, a therapeutic drug that is given to Neonates to improve lung capacity and where it would be required to monitor for toxicity risk. Caffeine is a listed analyte that is added gravimetrically, and this has a clinical function as the primary treatment of the breathing disorders apnea of prematurity. Another important population is with any patient that has liver failure. It is important if the laboratory tests hyperbilirubinemia patient samples that they analyze QC regularly to monitor this range to ensure accuracy and precision at these elevated levels. The requirement for a high concentration of Bilirubin presents a conflict for consolidation of multi-analytes. This means that not only is a separate paediatric control required, but a control matrix that is treated with antioxidants to inhibit the oxidation of Bilirubin. The application of the most appropriate measures to reduce the effect of oxidation on the high levels of Bilirubin in the Control have been researched to ensure that open vial and closed stability is maintained. Multichem NB Control is designed as a single level, liquid stable frozen product. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem P (P for protein), from Technopath Manufacturing Ltd, is specifically designed as a supplementary QC product for immunoproteins to be used in conjunction with the Multichem clinical chemistry QC. This product offers elevated concentrations for immunoprotein. Users can use this in conjunction with 1, 2 or 3 levels of Multichem S Plus or Multichem S, each of which already include multiple concentrations for immunoproteins. The full list of products available are shown below. Multichem P Supplementary Immunoprotein Control is intended for use as a third party, single level, liquid stable immunoprotein quality control material to monitor the precision of laboratory testing procedures for Immunoprotein Assays. Target values are provided for 39 analytes, including immunoglobulins, complement proteins, inflammatory proteins as well as carrier and storage proteins. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem P (P for protein), from Technopath Manufacturing Ltd, is specifically designed as a supplementary QC product for immunoproteins to be used in conjunction with the Multichem clinical chemistry QC. This product offers elevated concentrations for immunoprotein. Users can use this in conjunction with 1, 2 or 3 levels of Multichem S Plus or Multichem S, each of which already include multiple concentrations for immunoproteins. The full list of products available are shown below. Multichem P Supplementary Immunoprotein Control is intended for use as a third party, single level, liquid stable immunoprotein quality control material to monitor the precision of laboratory testing procedures for Immunoprotein Assays. Target values are provided for 39 analytes, including immunoglobulins, complement proteins, inflammatory proteins as well as carrier and storage proteins. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem S Plus Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Chemistry Assays. It incorporates serum chemistry, immunology, lipid, TDM, enzymes and esoterics. Multichem S Plus contains 105 analytes including the addition of C-Reactive Protein and Rheumatoid Factor at clinically relevant concentrations. Chemistry, Esoterics, Immunoproteins, Enzymes, Lipids, and Therapeutic Drugs are also included. When combined with Multichem P the solution offers an extensive spread of immunoprotein concentrations. Multichem S is also available, please note that Multichem S contains all the same analytes apart from C-Reactive Protein and Rhematoid Factor. Multichem S Plus is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem S Plus Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Chemistry Assays. It incorporates serum chemistry, immunology, lipid, TDM, enzymes and esoterics. Multichem S Plus contains 105 analytes including the addition of C-Reactive Protein and Rheumatoid Factor at clinically relevant concentrations. Chemistry, Esoterics, Immunoproteins, Enzymes, Lipids, and Therapeutic Drugs are also included. When combined with Multichem P the solution offers an extensive spread of immunoprotein concentrations. Multichem S is also available, please note that Multichem S contains all the same analytes apart from C-Reactive Protein and Rhematoid Factor. Multichem S Plus is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -
 Multichem U Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable, multi-analyte control designed for use in the routine monitoring of precision of laboratory testing procedures. Target values are provided for 13 analytes including kidney function parameters, pituitary/adrenal metabolites, hCG and urinary proteins. Multichem U Control is designed as a bi-level liquid stable product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at 2°C to 8°C and mixed as per IFU prior to use. The control is to be stored at 2 to 8°C between use.
Multichem U Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable, multi-analyte control designed for use in the routine monitoring of precision of laboratory testing procedures. Target values are provided for 13 analytes including kidney function parameters, pituitary/adrenal metabolites, hCG and urinary proteins. Multichem U Control is designed as a bi-level liquid stable product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at 2°C to 8°C and mixed as per IFU prior to use. The control is to be stored at 2 to 8°C between use. -
 Multichem WBT Control, from Technopath Manufacturing Ltd, is intended for use as a third-party, tri- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Cyclosporine, Tacrolimus and Sirolimus Assays. Multichem WBT Control is designed as a tri-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem WBT Control, from Technopath Manufacturing Ltd, is intended for use as a third-party, tri- level, liquid stable quality control material to monitor the precision of laboratory testing procedures for Cyclosporine, Tacrolimus and Sirolimus Assays. Multichem WBT Control is designed as a tri-level, liquid stable frozen product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.







