-

 Enteral Drainage Bags to fit any ENFit compliant tubes used for gastric drainage EVA sizes 50ml - 1000ml & PVC sizes 500ml - 2000ml to cover full range of patient requirements. The 500ml and 1000ml bags include integral tubing.
Enteral Drainage Bags to fit any ENFit compliant tubes used for gastric drainage EVA sizes 50ml - 1000ml & PVC sizes 500ml - 2000ml to cover full range of patient requirements. The 500ml and 1000ml bags include integral tubing. -

 Extension set/syringe driver connection set, male ENFit connector (ISO 80369-3) and female ENFit connector.
Extension set/syringe driver connection set, male ENFit connector (ISO 80369-3) and female ENFit connector. -

 Enteral ISO-SAF Home Enteral ISO SAF Home, reusable enteral syringe with female ENFit connection (ISO 80369-3). Low dose connections are fitted to the 1ml and 3ml syringes.
Enteral ISO-SAF Home Enteral ISO SAF Home, reusable enteral syringe with female ENFit connection (ISO 80369-3). Low dose connections are fitted to the 1ml and 3ml syringes.- 1ml
- 3ml
- 5ml
- 10ml.
- 20ml
- 60ml
-

 Enteral ISO-SAF Single Use Enteral ISO SAF, single use enteral syringes with female ENFit connection (ISO 80369-3). Low dose option available in 1ml & 3ml syringes.
Enteral ISO-SAF Single Use Enteral ISO SAF, single use enteral syringes with female ENFit connection (ISO 80369-3). Low dose option available in 1ml & 3ml syringes.- 1ml
- 3ml
- 5ml
- 10ml.
- 20ml
- 60ml
-

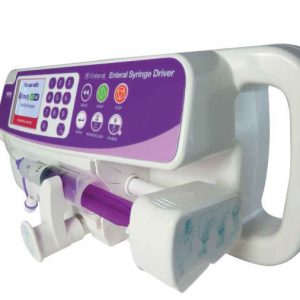 Enteral Syringe Driver For the application of enteral nutrition only. Deliver small volumes of enteral feed, accurately and safely, using enteral ENFit syringes.
Enteral Syringe Driver For the application of enteral nutrition only. Deliver small volumes of enteral feed, accurately and safely, using enteral ENFit syringes. -

 Hands-on training & performance verification improves pathogen detection. Data collected from environmental sampling of surfaces is highly dependent on the operator. Understanding and quantitatively characterising the percent recovery and consistency of sample collection by an operator drives improved detection and prevention of pathogens. Performance verification involves determining the percent recovery and consistency of an operator who collects samples in an environmental sampling program. Introducing Enverify™, a quantitative positive control for environmental sampling programs and comprises a flat substrate with a specific quantity of viable microorganisms coated on the surface. The microbes are safe-to-handle and are coated across a 10 cm x 10 cm area. Enverify™ provides a standardised tool for hands-on training and performance verification of operators that collect samples in environmental sampling programs. Enverify™ is compatible with contact plates, swab sampling devices, and sponge sample devices. Activities Involving Environmental Sampling:
Hands-on training & performance verification improves pathogen detection. Data collected from environmental sampling of surfaces is highly dependent on the operator. Understanding and quantitatively characterising the percent recovery and consistency of sample collection by an operator drives improved detection and prevention of pathogens. Performance verification involves determining the percent recovery and consistency of an operator who collects samples in an environmental sampling program. Introducing Enverify™, a quantitative positive control for environmental sampling programs and comprises a flat substrate with a specific quantity of viable microorganisms coated on the surface. The microbes are safe-to-handle and are coated across a 10 cm x 10 cm area. Enverify™ provides a standardised tool for hands-on training and performance verification of operators that collect samples in environmental sampling programs. Enverify™ is compatible with contact plates, swab sampling devices, and sponge sample devices. Activities Involving Environmental Sampling:- Environmental monitoring for pathogens or indicator organisms
- Sanitation verification using ATP or indicator organisms
- Verification of hygienic restoration following adverse events
- Verification of containment during construction activities
-

 Microbial-coated test surfaces for evaluating surface sampling competency Qualification of personnel who collect surface samples is a regulatory requirement and a crucial activity for an effective environmental monitoring program. Personnel sampling performance, including percent recovery, consistency, and aseptic technique, drives the quality of data that is generated and significantly impacts the integrity of an environmental monitoring program. Introducing Enverify™, the industry standard for evaluating surface sampling competency. Enverify™ Test Surfaces, from Stratix Labs, are the first standardised tool that enables you to evaluate and document surface sampling competency with quantitative data. Each Enverify™ Test Surface contains a precise quantity of microorganisms, providing a benchmark for you to evaluate, verify, and compare performance. Enverify™ is designed for use with contact plates and swab sampling tools.
Microbial-coated test surfaces for evaluating surface sampling competency Qualification of personnel who collect surface samples is a regulatory requirement and a crucial activity for an effective environmental monitoring program. Personnel sampling performance, including percent recovery, consistency, and aseptic technique, drives the quality of data that is generated and significantly impacts the integrity of an environmental monitoring program. Introducing Enverify™, the industry standard for evaluating surface sampling competency. Enverify™ Test Surfaces, from Stratix Labs, are the first standardised tool that enables you to evaluate and document surface sampling competency with quantitative data. Each Enverify™ Test Surface contains a precise quantity of microorganisms, providing a benchmark for you to evaluate, verify, and compare performance. Enverify™ is designed for use with contact plates and swab sampling tools. -

 Environmental testing and water quality control are a major concern for public health and are subject to laws and regulations in many countries. Water-bearing systems contaminated with pathogens are a source of infection and must be tested. BIOTECON Diagnostics’ real-time PCR-based microproof® LyoKits play a trusted role in detection and quantification of pathogens in environmental samples and water, and offer reliable analysis with high sensitivity and specificity.
Environmental testing and water quality control are a major concern for public health and are subject to laws and regulations in many countries. Water-bearing systems contaminated with pathogens are a source of infection and must be tested. BIOTECON Diagnostics’ real-time PCR-based microproof® LyoKits play a trusted role in detection and quantification of pathogens in environmental samples and water, and offer reliable analysis with high sensitivity and specificity. -

 Enteral Feeding Tube Extension Sets Mini Compact Feeding Sets (Extension sets) are available in various configurations. e.g: 2″ Straight Feeding Sets/Medication set are useful for flushing, right angle sets are preferred for feeding via a pump versus straight sets are more beneficial for bolus feeding.
Enteral Feeding Tube Extension Sets Mini Compact Feeding Sets (Extension sets) are available in various configurations. e.g: 2″ Straight Feeding Sets/Medication set are useful for flushing, right angle sets are preferred for feeding via a pump versus straight sets are more beneficial for bolus feeding. -

 A pathogen is a synonym for a disease-causing microorganism. Food pathogens represent a special form of microbial pathogens, which are picked up and spread through food. Below a number of pathogens are listed, for which BIOTECON Diagnostics offers suitable foodproof® DNA extraction and real-time PCR detection kits. Many of the pathogen kits have been certified by recognized institutions such as AOAC-RI, MicroVal or NordVal (see licensing options / validations), and thus independently tested for quality and safety.
A pathogen is a synonym for a disease-causing microorganism. Food pathogens represent a special form of microbial pathogens, which are picked up and spread through food. Below a number of pathogens are listed, for which BIOTECON Diagnostics offers suitable foodproof® DNA extraction and real-time PCR detection kits. Many of the pathogen kits have been certified by recognized institutions such as AOAC-RI, MicroVal or NordVal (see licensing options / validations), and thus independently tested for quality and safety. -

 Foodborne viruses can cause various, sometimes serious conditions. Contamination of food and bottled water is usually through infected persons or contaminated waste water. Viruses can not reproduce on food or in water, but small amounts of residing viruses may be sufficient to cause infection. The standard method for virus detection in food and water samples is PCR. BIOTECON Diagnostics offers a Hepatitis A virus kit, a norovirus real-time PCR kit and a new multiplex system for both virus particles specifically for the investigation of food. All kits contain a ready-to-use Process Control solution. With each sample you can detect a Process Control (MS2-phage in our case).
Foodborne viruses can cause various, sometimes serious conditions. Contamination of food and bottled water is usually through infected persons or contaminated waste water. Viruses can not reproduce on food or in water, but small amounts of residing viruses may be sufficient to cause infection. The standard method for virus detection in food and water samples is PCR. BIOTECON Diagnostics offers a Hepatitis A virus kit, a norovirus real-time PCR kit and a new multiplex system for both virus particles specifically for the investigation of food. All kits contain a ready-to-use Process Control solution. With each sample you can detect a Process Control (MS2-phage in our case). -

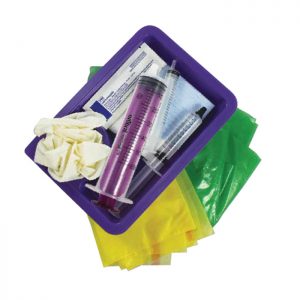 G-Pak The pack contains everything you need for a Gastrostomy change in one easy-to-use pack. Pack is contained in a Stericrepe Paper 60 x 60cm wrapper and contains:
G-Pak The pack contains everything you need for a Gastrostomy change in one easy-to-use pack. Pack is contained in a Stericrepe Paper 60 x 60cm wrapper and contains:- 2 x Gloves - Latex Free
- 1 x Plastic Apron
- 1 x Lubrication Aqua Gel
- 1 x 5 Swab, Plain, Gauze 10 x 10cm, 12-ply White
- 1 x Syringe 10ml Sterile Water Prefilled
- 1 x Syringe, 10ml Luer Slip Eccentric
- 1 x Syringe, 60ml ENFit ISO 80369-3
- 1 x Tray Gallipot
- 1 x Waste Bag
-

 Used for third degree prolapse and procidentia The Silicone Gellhorn Long Stem Pessary is used for a second to third degree uterine prolapse or procidentia.
Used for third degree prolapse and procidentia The Silicone Gellhorn Long Stem Pessary is used for a second to third degree uterine prolapse or procidentia. -

 Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia.
Used for third degree prolapse and procidentia The Silicone Gellhorn Short Stem Pessary is used for a second to third degree uterine prolapse or procidentia. -

 Enabling Quick Intubations Specially designed to work with GlideScope® video laryngoscopes, GlideRite® Stylets enable efficient endotracheal tube placement. GlideRite® Single-use Stylet - Small Designed for use in ET tubes 3.0 mm to 4.0 mm GlideRite® Single-use Stylet - Large Designed for use in ET tubes 6.0 mm and larger GlideRite® Rigid Stylet Designed for use in ET tubes 6.0 mm and larger GlideRite® DLT Stylet Designed for use in double lumen ET tubes 6.0 mm and larger
Enabling Quick Intubations Specially designed to work with GlideScope® video laryngoscopes, GlideRite® Stylets enable efficient endotracheal tube placement. GlideRite® Single-use Stylet - Small Designed for use in ET tubes 3.0 mm to 4.0 mm GlideRite® Single-use Stylet - Large Designed for use in ET tubes 6.0 mm and larger GlideRite® Rigid Stylet Designed for use in ET tubes 6.0 mm and larger GlideRite® DLT Stylet Designed for use in double lumen ET tubes 6.0 mm and larger -

 Real-time Airway View The GlideScope® AVL video laryngoscope system delivers a consistently clear, real-time view of the airway. Get a better airway view with a large, full colour monitor. Improve training and document intubation results with features such as snapshot and video recording. 6 Single-use sizes available. The AVL system offers 6 single-use GVL Stat sizes for patients from neonate to large adult.
Real-time Airway View The GlideScope® AVL video laryngoscope system delivers a consistently clear, real-time view of the airway. Get a better airway view with a large, full colour monitor. Improve training and document intubation results with features such as snapshot and video recording. 6 Single-use sizes available. The AVL system offers 6 single-use GVL Stat sizes for patients from neonate to large adult. -

 A Brighter Vision in Single-use Bronchoscopy GlideScope® Bflex™ is a single-use bronchoscope designed with exceptional image quality and manoeuvrability for bronchoscopy procedures and difficult airways. BFlex single-use bronchoscopes combine the convenience and accessibility of single-use tools with the performance you expect from a reusable bronchoscope. IMPROVE EFFICIENCY WHILE REDUCING COSTS Single-use bronchoscopes may provide better accessibility and cost effectiveness compared to reusable bronchoscopes.
A Brighter Vision in Single-use Bronchoscopy GlideScope® Bflex™ is a single-use bronchoscope designed with exceptional image quality and manoeuvrability for bronchoscopy procedures and difficult airways. BFlex single-use bronchoscopes combine the convenience and accessibility of single-use tools with the performance you expect from a reusable bronchoscope. IMPROVE EFFICIENCY WHILE REDUCING COSTS Single-use bronchoscopes may provide better accessibility and cost effectiveness compared to reusable bronchoscopes. -

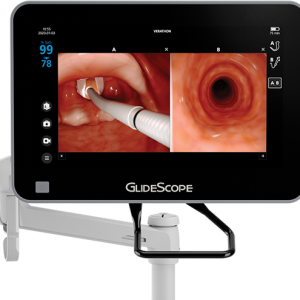 The power to see more GlideScope® Core™, the most comprehensive and flexible airway visualization system for video laryngoscopy, bronchoscopy and multimodal airway procedures. GlideScope Core is an all-in-one system offering immediate access to the tools you need to visualize the airway and tracheobronchial tree. Designed around a high-definition, touchscreen monitor and comprehensive workstation, GlideScope Core delivers elevated visibility and improved workflow. GlideScope Core is the first airway visualization system to offer live multimodal image capability, giving you the power to view the airway via a GlideScope video laryngoscope and BFlex™ single-use flexible bronchoscope – simultaneously. Choose a 10” OR 15” Touchscreen monitor.
The power to see more GlideScope® Core™, the most comprehensive and flexible airway visualization system for video laryngoscopy, bronchoscopy and multimodal airway procedures. GlideScope Core is an all-in-one system offering immediate access to the tools you need to visualize the airway and tracheobronchial tree. Designed around a high-definition, touchscreen monitor and comprehensive workstation, GlideScope Core delivers elevated visibility and improved workflow. GlideScope Core is the first airway visualization system to offer live multimodal image capability, giving you the power to view the airway via a GlideScope video laryngoscope and BFlex™ single-use flexible bronchoscope – simultaneously. Choose a 10” OR 15” Touchscreen monitor. -

 Handheld System - Portability without Compromise Verathon’s second generation handheld system delivering the confidence, clarity and ease-of-use of GlideScope®, your trusted brand of video laryngoscopes designed for rapid, first pass success1. GlideScope Go 2 is IEC 60601-1-12:2014 certified for use in Emergency Medical Services (EMS) environments. Get the confidence of GlideScope® whenever and wherever you need it, with the portable GlideScope® Go™ — the new handheld system created for both hospital and pre-hospital settings. Intubate routine and difficult airways with the handheld system, designed for 1st pass success.
Handheld System - Portability without Compromise Verathon’s second generation handheld system delivering the confidence, clarity and ease-of-use of GlideScope®, your trusted brand of video laryngoscopes designed for rapid, first pass success1. GlideScope Go 2 is IEC 60601-1-12:2014 certified for use in Emergency Medical Services (EMS) environments. Get the confidence of GlideScope® whenever and wherever you need it, with the portable GlideScope® Go™ — the new handheld system created for both hospital and pre-hospital settings. Intubate routine and difficult airways with the handheld system, designed for 1st pass success.1. Silverberg, Michael J., et al. “Comparison of Video Laryngoscopy Versus Direct Laryngoscopy During Urgent Endotracheal Intubation: A Randomized Controlled Trial.” Critical Care Medicine 43.3 (2014): 636-641.
-

 Designed for 1st Pass Success GlideScope® Spectrum™ Single-use video laryngoscopes combine fully disposable low profile blades with the latest advancements in lighting and camera technology. Compatible with the GlideScope® Go, GlideScope® Core and GlideScope® Video Monitor. With the Spectrum system, seven blade sizes enable quick intubations across a wide range of patient types, weights, and clinical settings. The LoPro blades offer the signature GlideScope blade angulation and cover a patient range from neonate to large adult. The DirectView MAC blades extend system flexibility, incorporating the look and feel of traditional MAC blades with the confidence of a GlideScope. Available in 7 sizes.
Designed for 1st Pass Success GlideScope® Spectrum™ Single-use video laryngoscopes combine fully disposable low profile blades with the latest advancements in lighting and camera technology. Compatible with the GlideScope® Go, GlideScope® Core and GlideScope® Video Monitor. With the Spectrum system, seven blade sizes enable quick intubations across a wide range of patient types, weights, and clinical settings. The LoPro blades offer the signature GlideScope blade angulation and cover a patient range from neonate to large adult. The DirectView MAC blades extend system flexibility, incorporating the look and feel of traditional MAC blades with the confidence of a GlideScope. Available in 7 sizes. -

 Lightweight titanium for added strength and durability Innovative construction and blade styles for a wide array of clinical demands. Incorporating an advanced digital camera with an integrated LED light source, GlideScope® Titanium™ laryngoscopes connect to the all new GlideScope® Core™ system for real time viewing and recording. and with a choice of low-profile blades in two distinct options, you can choose the way you want to work. Available in 5 sizes.
Lightweight titanium for added strength and durability Innovative construction and blade styles for a wide array of clinical demands. Incorporating an advanced digital camera with an integrated LED light source, GlideScope® Titanium™ laryngoscopes connect to the all new GlideScope® Core™ system for real time viewing and recording. and with a choice of low-profile blades in two distinct options, you can choose the way you want to work. Available in 5 sizes. -

 Reusable Video Baton GlideScope® Video Baton 2.0 Large is a reusable video baton that works with single-use stats to provide a consistently clear, real-time view of the airway. We are continually developing new devices and accessories to meet the demands of your changing environment. Our Video Baton 2.0 Large is compatible with GlideScope® Go™ and GlideScope® Core™, making it easier to use and to purchase products from a trusted single source.
Reusable Video Baton GlideScope® Video Baton 2.0 Large is a reusable video baton that works with single-use stats to provide a consistently clear, real-time view of the airway. We are continually developing new devices and accessories to meet the demands of your changing environment. Our Video Baton 2.0 Large is compatible with GlideScope® Go™ and GlideScope® Core™, making it easier to use and to purchase products from a trusted single source. -

 Genetically modified crops cover over 10% of global agricultural land. More than one hundred different genetically modified plants are grown on over 160 million hectares in more than 29 countries. The main producing countries are the USA, Brazil, Argentina, Canada and India. Soybeans are the most highly cultivated genetically modified plant, followed by corn, cotton and canola. According to EU Regulation 1829/2003 and 1830/2003, GMO content of more than 0.9% in foods and ingredients must be labeled. Other countries and regions have similar rules with similar limits. For reliable analysis of GMOs in food and animal feed, BIOTECON Diagnostics offers a broad range of solutions.
Genetically modified crops cover over 10% of global agricultural land. More than one hundred different genetically modified plants are grown on over 160 million hectares in more than 29 countries. The main producing countries are the USA, Brazil, Argentina, Canada and India. Soybeans are the most highly cultivated genetically modified plant, followed by corn, cotton and canola. According to EU Regulation 1829/2003 and 1830/2003, GMO content of more than 0.9% in foods and ingredients must be labeled. Other countries and regions have similar rules with similar limits. For reliable analysis of GMOs in food and animal feed, BIOTECON Diagnostics offers a broad range of solutions. -

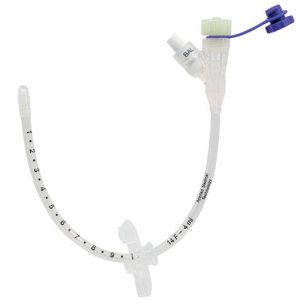 AMT Balloon G-Tube The AMT Balloon G-Tube is a Silicone replacement gastrostomy tube (G-Tube). The G-Tube is placed through the abdominal wall to give direct access to the stomach and has an adjustable external bolster minimising surface contact with the skin, allowing the tissue to breathe and can be adjusted up to 10cm in length.
AMT Balloon G-Tube The AMT Balloon G-Tube is a Silicone replacement gastrostomy tube (G-Tube). The G-Tube is placed through the abdominal wall to give direct access to the stomach and has an adjustable external bolster minimising surface contact with the skin, allowing the tissue to breathe and can be adjusted up to 10cm in length.



