-

 Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms.
Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms. -

 Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program
Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program -
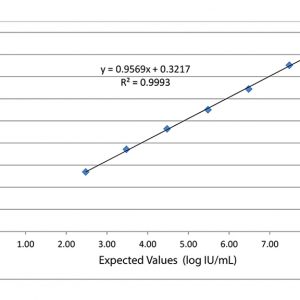
 Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration.
Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration. -

 Available for use with serological and molecular assays, SeraCare’s portfolio of AccuSet performance panels contains highly characterized, raw, undiluted plasma specimens collected from unique individuals positive for your analytes of interest. Each panel contains a comprehensive comparative data sheet with test results from a wide variety of leading commercially available assays and platforms. AccuSet performance panels can be used to evaluate assay specificity, sensitivity, repeatability, and reproducibility to assist you in validating new test methods and equipment, run head-to-head assay comparisons, demonstrate lab proficiency, and train laboratory personnel.
Available for use with serological and molecular assays, SeraCare’s portfolio of AccuSet performance panels contains highly characterized, raw, undiluted plasma specimens collected from unique individuals positive for your analytes of interest. Each panel contains a comprehensive comparative data sheet with test results from a wide variety of leading commercially available assays and platforms. AccuSet performance panels can be used to evaluate assay specificity, sensitivity, repeatability, and reproducibility to assist you in validating new test methods and equipment, run head-to-head assay comparisons, demonstrate lab proficiency, and train laboratory personnel. -
 Multichem U Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable, multi-analyte control designed for use in the routine monitoring of precision of laboratory testing procedures. Target values are provided for 13 analytes including kidney function parameters, pituitary/adrenal metabolites, hCG and urinary proteins. Multichem U Control is designed as a bi-level liquid stable product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at 2°C to 8°C and mixed as per IFU prior to use. The control is to be stored at 2 to 8°C between use.
Multichem U Control, from Technopath Manufacturing Ltd, is intended for use as a third party, bi- level, liquid stable, multi-analyte control designed for use in the routine monitoring of precision of laboratory testing procedures. Target values are provided for 13 analytes including kidney function parameters, pituitary/adrenal metabolites, hCG and urinary proteins. Multichem U Control is designed as a bi-level liquid stable product. The product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at 2°C to 8°C and mixed as per IFU prior to use. The control is to be stored at 2 to 8°C between use. -
 Multichem P (P for protein), from Technopath Manufacturing Ltd, is specifically designed as a supplementary QC product for immunoproteins to be used in conjunction with the Multichem clinical chemistry QC. This product offers elevated concentrations for immunoprotein. Users can use this in conjunction with 1, 2 or 3 levels of Multichem S Plus or Multichem S, each of which already include multiple concentrations for immunoproteins. The full list of products available are shown below. Multichem P Supplementary Immunoprotein Control is intended for use as a third party, single level, liquid stable immunoprotein quality control material to monitor the precision of laboratory testing procedures for Immunoprotein Assays. Target values are provided for 39 analytes, including immunoglobulins, complement proteins, inflammatory proteins as well as carrier and storage proteins. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem P (P for protein), from Technopath Manufacturing Ltd, is specifically designed as a supplementary QC product for immunoproteins to be used in conjunction with the Multichem clinical chemistry QC. This product offers elevated concentrations for immunoprotein. Users can use this in conjunction with 1, 2 or 3 levels of Multichem S Plus or Multichem S, each of which already include multiple concentrations for immunoproteins. The full list of products available are shown below. Multichem P Supplementary Immunoprotein Control is intended for use as a third party, single level, liquid stable immunoprotein quality control material to monitor the precision of laboratory testing procedures for Immunoprotein Assays. Target values are provided for 39 analytes, including immunoglobulins, complement proteins, inflammatory proteins as well as carrier and storage proteins. The product will typically be run as a supplementary high level control, in conjunction with at least one of the Multichem Serum Control levels, (S or S Plus) to provide a minimum of 2 levels of control to allow assay system performance monitoring within the analytical ranges. It will be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -
 Multichem S Plus Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Chemistry Assays. It incorporates serum chemistry, immunology, lipid, TDM, enzymes and esoterics. Multichem S Plus contains 105 analytes including the addition of C-Reactive Protein and Rheumatoid Factor at clinically relevant concentrations. Chemistry, Esoterics, Immunoproteins, Enzymes, Lipids, and Therapeutic Drugs are also included. When combined with Multichem P the solution offers an extensive spread of immunoprotein concentrations. Multichem S is also available, please note that Multichem S contains all the same analytes apart from C-Reactive Protein and Rhematoid Factor. Multichem S Plus is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem S Plus Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Chemistry Assays. It incorporates serum chemistry, immunology, lipid, TDM, enzymes and esoterics. Multichem S Plus contains 105 analytes including the addition of C-Reactive Protein and Rheumatoid Factor at clinically relevant concentrations. Chemistry, Esoterics, Immunoproteins, Enzymes, Lipids, and Therapeutic Drugs are also included. When combined with Multichem P the solution offers an extensive spread of immunoprotein concentrations. Multichem S is also available, please note that Multichem S contains all the same analytes apart from C-Reactive Protein and Rhematoid Factor. Multichem S Plus is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -

 Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA Plus Control is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Plus contains 86 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers, bone metabolism markers and tumour markers. Multichem IA Plus, from Technopath Manufacturing Ltd, is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -

 Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use.
Multichem IA contains 83 analytes including fertility and thyroid hormones, steroid hormones, cardiac markers, anaemia markers, therapeutic drugs, adrenal markers and bone metabolism markers. Multichem IA Control, from Technopath Manufacturing Ltd, is intended for use as a third party, multi-constituent quality control material to monitor the precision of laboratory testing procedures for Immunoassay Assays. Please note: the main difference between Multichem IA Plus and Multichem IA product is the addition of three tumor markers to Multichem IA Plus; CA 125, CA 15-3 and CA 19-9. Multichem IA is designed as a tri-level, liquid stable frozen product. This product will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control should be stored at -20°C to –80°C and thawed as per IFU prior to use. The product should be stored at 2 to 8°C between use. -
 Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use.
Multichem hsTn Control, from Technopath Manufacturing Ltd, is intended for use as a third party, single level, liquid stable quality control material to monitor the precision of laboratory testing procedures for high sensitive Troponin Assays. The intended patient population is Cardiac patients, where a requirement has been identified for testing conditions associated with elevated levels of Troponin I and Troponin T. Multichem hsTn Control is designed as a single level, liquid stable frozen product. The product will typically be run as a low level/high sensitive control, in conjunction with one other level of the Multichem IA / Multichem IA Plus series of serum controls for the same analytes, after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. This product design is based on the existing Multichem IA Plus L1 control, but incorporating the required features of a single low level Troponin Control, to supplement the IA Plus Control to monitor the precision of Troponin assay methods, at the low end of the linear dynamic range. The product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The control is to be stored at -20°C to –80°C and thawed as per IFU prior to use. The product is to be stored at 2 to 8°C between use. -

 Your perfect tool to aid in the monitoring of instrument detection of Haemolysis, Icterus and Lipaemia in specimens. Fortress Serum Indices Quality Control are designed to monitor the ability of an instrument to accurately measure HIL in specimens. This is done through the manufacture of control material which mimics specimens that would be considered normal, haemolytic, icteric, and lipaemic using human based products. Ensure accurate monitoring of patient samples for Serum Indices by running this QC daily.
Your perfect tool to aid in the monitoring of instrument detection of Haemolysis, Icterus and Lipaemia in specimens. Fortress Serum Indices Quality Control are designed to monitor the ability of an instrument to accurately measure HIL in specimens. This is done through the manufacture of control material which mimics specimens that would be considered normal, haemolytic, icteric, and lipaemic using human based products. Ensure accurate monitoring of patient samples for Serum Indices by running this QC daily. -
 Multichem IA Speciality Immunoassay Controls are intended for use as third party, tri- level, liquid stable, multi-analyte quality control materials to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Speciality, from Technopath Manufacturing Ltd, is designed to complement Multichem IA and Multichem IA Plus Immunoassay Controls by offering significantly increased stability for BNP, PTH and ACTH. In addition to these analytes, Multichem IA Speciality Controls also contains and provides tri level utility for Procalcitonin and Calcitonin. Multichem IA Speciality Immunoassay Controls are designed as tri-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls should be stored at -20°C to –80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use.
Multichem IA Speciality Immunoassay Controls are intended for use as third party, tri- level, liquid stable, multi-analyte quality control materials to monitor the precision of laboratory testing procedures for Immunoassay Assays. Multichem IA Speciality, from Technopath Manufacturing Ltd, is designed to complement Multichem IA and Multichem IA Plus Immunoassay Controls by offering significantly increased stability for BNP, PTH and ACTH. In addition to these analytes, Multichem IA Speciality Controls also contains and provides tri level utility for Procalcitonin and Calcitonin. Multichem IA Speciality Immunoassay Controls are designed as tri-level, liquid stable frozen products. These products will typically be run after reagent calibration and at a frequency dictated by laboratory QC procedures and reagent / instrument manufacturer instructions. The products should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent being used. The controls should be stored at -20°C to –80°C and thawed as per IFU prior to use. The products should be stored at 2 to 8°C between use.



