-

 Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing.
Ensure antibody testing accuracy and performance AccuSet™ SARS-CoV-2 Performance Panel from SeraCare - This eleven-member validation panel consists of undiluted, naturally occurring human-plasma members. Ten panel members represent collections from multiple individuals positive for antibodies to SARS-CoV-2; a single negative member is also included. Test results from commercially available SARS-CoV-2 antibody assays are included for comparative analysis. AccuSet™ SARS-CoV-2 Performance Panel is intended to provide an out-of-the-box solution to evaluate SARS-CoV-2 antibody detection assays with highly characterized human specimens whether generating validation data for a regulatory submission or performing assay verification in a clinical laboratory setting. ACCURUN® Anti-SARS-CoV-2 Reference Material Kit is designed to support assay installation and monitoring of day-to-day assay performance, providing a complete quality solution for SARS-CoV-2 antibody testing. -
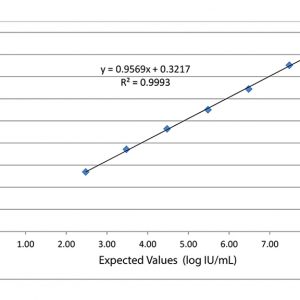
 Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration.
Validate and Monitor your assays AccuSpan linearity panels from SeraCare are designed to span the dynamic range of quantitative infectious disease assays and evaluate instrumentation analytical sensitivity. Linearity panels effectively challenge assay performance at defined intervals to ensure consistency throughout the entire reportable range. In addition to linearity studies, these panels are useful in validation procedures for new assay implementation, operator training, and troubleshooting signs of assay deterioration. -

 Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program
Ensure reagents are operating effectively from lot-to-lot AccuTrak qualification panels from SeraCare are designed as a cost-effective solution to deliver the consistent results you need to gain confidence in your assay’s performance and ensure reagents are operating effectively from lot-to-lot. SeraCare's AccuTrak qualification panels are utilised by clinical laboratories worldwide to help strengthen quality control protocols and procedures for infectious disease diagnostic assays. With products for HIV, hepatitis, CMV, syphilis, HPV, and HTLV, SeraCare offers a comprehensive portfolio to help effectively monitor assay performance. Reliable, Consistent, Cost-Effective Solutions for Your Assay QC Program -

 Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms.
Assess your assay development milestones When your assay development requires natural patient specimens that represent the body’s true response to an infection, you can depend on AccuVert seroconversion panels as a gold standard with which to assess your assay development milestones. The SeraCare seroconversion panels are developed using raw, undiluted plasma collected from a single individual during the development of an infection and subsequent immunological response. Spanning an array of infectious diseases from HIV to hepatitis and syphilis, SeraCare's panels provide you with a diverse selection of products with high-quality datasets to help evaluate your assay. A Rich History in Seroconversion Panels SeraCare have been a trusted provider of seroconversion panels for over 30 years, IVD diagnostic manufacturers worldwide have used our panels in the development and validation of their assays for decades. As shown in the World Health Organisation HIV test evaluation kit reports, SeraCare HIV seroconversion panels have been used for comparative studies for HIV test kit evaluations, and are also frequently referenced in package inserts of leading IVD infectious disease platforms. -

 Revolutionising Histopathology with Fully Automated Microtomy Automated microtomy is transforming the way histopathology laboratories operate—improving consistency, throughput, and diagnostic turnaround times. The Morphle Robotome (RTM30) represents a breakthrough in robotic microtomy and slide preparation, enabling laboratories to overcome traditional bottlenecks in tissue sectioning. Designed for modern cytology, histopathology, and cancer diagnostics laboratories, the Robotome automates the critical sectioning process—delivering high-quality, reproducible slides at scale, while reducing manual workload and variability. Key Applications: The Morphle Robotome is ideally suited for:
Revolutionising Histopathology with Fully Automated Microtomy Automated microtomy is transforming the way histopathology laboratories operate—improving consistency, throughput, and diagnostic turnaround times. The Morphle Robotome (RTM30) represents a breakthrough in robotic microtomy and slide preparation, enabling laboratories to overcome traditional bottlenecks in tissue sectioning. Designed for modern cytology, histopathology, and cancer diagnostics laboratories, the Robotome automates the critical sectioning process—delivering high-quality, reproducible slides at scale, while reducing manual workload and variability. Key Applications: The Morphle Robotome is ideally suited for:- Histopathology laboratories
- Cancer diagnostics centres
- Cytology workflows
- Biopharma and translational research labs
- High-volume diagnostic services
-

 Auto Haematology Analyser with ESR BC-700 Series is a revolutionary haematology analyser series that incorporates both complete blood count (CBC) and erythrocyte sedimentation rate (ESR) tests. This series, including two open vial models BC-700/BC-720 and two autoloader models BC-760/BC-780, is designed to empower medium-volume laboratories with advanced diagnostics technologies that are applied in the premium products. In addition, it saves the costs that would otherwise be incurred for the purchase, maintenance, consumables, and storage space of a separate ESR analyser. Compared with the traditional Westergren method, this method performs better in quality traceability, repeatability, speed, safety, and level of automation.
Auto Haematology Analyser with ESR BC-700 Series is a revolutionary haematology analyser series that incorporates both complete blood count (CBC) and erythrocyte sedimentation rate (ESR) tests. This series, including two open vial models BC-700/BC-720 and two autoloader models BC-760/BC-780, is designed to empower medium-volume laboratories with advanced diagnostics technologies that are applied in the premium products. In addition, it saves the costs that would otherwise be incurred for the purchase, maintenance, consumables, and storage space of a separate ESR analyser. Compared with the traditional Westergren method, this method performs better in quality traceability, repeatability, speed, safety, and level of automation. -

 Advancing Haematology with Automated Digital Cell Morphology The evolution of haematology laboratories is being driven by the need for faster, more accurate, and standardised blood film analysis. The Mindray MC-80 Automated Digital Cell Morphology Analyser brings together high-resolution imaging, AI-powered classification, and workflow automation to transform cellular analysis. Designed for modern haematology laboratories, cancer diagnostics, and clinical pathology services, the MC-80 enables rapid, reliable morphology review—reducing manual workload while improving diagnostic confidence. Key Applications: The Mindray MC-80 is ideal for:
Advancing Haematology with Automated Digital Cell Morphology The evolution of haematology laboratories is being driven by the need for faster, more accurate, and standardised blood film analysis. The Mindray MC-80 Automated Digital Cell Morphology Analyser brings together high-resolution imaging, AI-powered classification, and workflow automation to transform cellular analysis. Designed for modern haematology laboratories, cancer diagnostics, and clinical pathology services, the MC-80 enables rapid, reliable morphology review—reducing manual workload while improving diagnostic confidence. Key Applications: The Mindray MC-80 is ideal for:- Hospital haematology laboratories
- Cancer and oncology diagnostics centres
- Clinical pathology laboratories
- Reference and high-throughput diagnostic services
- Research and academic institutions
- Histopathology laboratories
-

 Internal Quality Control Software from Technopath Manufacturing Ltd. IAMQC Daily is a comprehensive Internal Quality Control software that applies Westgard and/or any user-defined QC rules to individual QC results. The software automatically builds interactive Levey-Jennings charts and tables and provides summary and detailed customised reports to the end user. IAMQC Daily integrates with Microsoft Excel to produce customised electronic reports. The system also allows the import of pre-defined templates, resulting in instant system setups. Users can create audit trails, action logs and summary reports at the click of a button. IAMQC Daily comprises a centralised program that facilitates the analysis of multiple QC materials, across numerous departments in a laboratory setting. The system can also be integrated with IAMQC Peer to satisfy both Internal and External QC requirements. IAMQC Daily offers a centralised review of all QC data from all laboratories/instruments. Central administrator access facilitates managers to review QC performance at multiple facilities – no need to visit each laboratory site. Closer monitoring of QC from remote locations without additional costs provides a flexible option for managing the inter and intra-lab performance. The software works on an ‘open’ platform that allows the end user to add all types of control material from a range of laboratories. Both IAMQC Daily and Expert can run using an internet OR intranet connection. Each PC license allows the user to manage an unlimited number of departments, control materials (not limited to Technopath materials) and instruments. An unlimited number of user logins can be added to the system at any stage. An administrator module can also manage user logins, customising the functionality that is available to each user. All data can also be filtered using the same logic to apply a user-friendly atmosphere and save time scrolling through data.
Internal Quality Control Software from Technopath Manufacturing Ltd. IAMQC Daily is a comprehensive Internal Quality Control software that applies Westgard and/or any user-defined QC rules to individual QC results. The software automatically builds interactive Levey-Jennings charts and tables and provides summary and detailed customised reports to the end user. IAMQC Daily integrates with Microsoft Excel to produce customised electronic reports. The system also allows the import of pre-defined templates, resulting in instant system setups. Users can create audit trails, action logs and summary reports at the click of a button. IAMQC Daily comprises a centralised program that facilitates the analysis of multiple QC materials, across numerous departments in a laboratory setting. The system can also be integrated with IAMQC Peer to satisfy both Internal and External QC requirements. IAMQC Daily offers a centralised review of all QC data from all laboratories/instruments. Central administrator access facilitates managers to review QC performance at multiple facilities – no need to visit each laboratory site. Closer monitoring of QC from remote locations without additional costs provides a flexible option for managing the inter and intra-lab performance. The software works on an ‘open’ platform that allows the end user to add all types of control material from a range of laboratories. Both IAMQC Daily and Expert can run using an internet OR intranet connection. Each PC license allows the user to manage an unlimited number of departments, control materials (not limited to Technopath materials) and instruments. An unlimited number of user logins can be added to the system at any stage. An administrator module can also manage user logins, customising the functionality that is available to each user. All data can also be filtered using the same logic to apply a user-friendly atmosphere and save time scrolling through data. -

 Select QC Rules and make meaningful QC decisions IAMQC Expert, from Technopath Manufacturing Ltd, is an interactive system that helps front line laboratory staff select QC rules, reduce unnecessary repeats and make meaningful QC decisions. IAMQC Expert allows the end-user to monitor method performance relative to clinical requirements and focus on the tests that require their attention. Times and technologies are changing rapidly. Instruments and methodologies are more accurate, precise and stable than they were a decade ago. Most laboratories have adopted these new technical advances, but few have modified their QC processes to match. Many laboratories are still using a 1-2s rule as recommended by Levey and Jennings in 1951. Technopath Manufacturing Ltd have designed a QC system that will alert users to significant changes and not generate QC flags when the system is operating safely within acceptable limits. The system compares method performance to defined quality requirements (rather than to last month’s data) and recommends QC strategies that will warn users when QC data points exceed acceptable performance - with a minimal number of false flags. In the design of our QC system we “balance” the quality control system to meet the changing performance and stability of the analytical system.
Select QC Rules and make meaningful QC decisions IAMQC Expert, from Technopath Manufacturing Ltd, is an interactive system that helps front line laboratory staff select QC rules, reduce unnecessary repeats and make meaningful QC decisions. IAMQC Expert allows the end-user to monitor method performance relative to clinical requirements and focus on the tests that require their attention. Times and technologies are changing rapidly. Instruments and methodologies are more accurate, precise and stable than they were a decade ago. Most laboratories have adopted these new technical advances, but few have modified their QC processes to match. Many laboratories are still using a 1-2s rule as recommended by Levey and Jennings in 1951. Technopath Manufacturing Ltd have designed a QC system that will alert users to significant changes and not generate QC flags when the system is operating safely within acceptable limits. The system compares method performance to defined quality requirements (rather than to last month’s data) and recommends QC strategies that will warn users when QC data points exceed acceptable performance - with a minimal number of false flags. In the design of our QC system we “balance” the quality control system to meet the changing performance and stability of the analytical system. -

 Innovative, real-time, Peer Comparison Software from Technopath Manufacturing Ltd. The web based system facilitates laboratories testing the same lot number of control material to access valuable information from their colleagues through peer comparison. The reports that are generated in IAMQC Peer compare the accuracy and precision of analytical processes between laboratories and peer groups. This information can be extremely valuable, indicating the user’s performance relative to their peer group and also providing powerful troubleshooting tools when attempting to resolve potential problems. To participate in IAMQC Peer, each individual laboratory submits their individual results or summary statistics (mean, standard deviation, and number of data points) to the central database maintained by Technopath Manufacturing Ltd. Laboratories data may be submitted manually on-line or, alternatively, captured by one of our many interfacing options. The information provided by IAMQC Peer can be used on a monthly basis to evaluate how well lab’s methods are operating, relative to the overall peer group. Users can also look at this peer data in real-time, interactive, tables online, when they are investigating a potential problem with accuracy or precision for an individual method.
Innovative, real-time, Peer Comparison Software from Technopath Manufacturing Ltd. The web based system facilitates laboratories testing the same lot number of control material to access valuable information from their colleagues through peer comparison. The reports that are generated in IAMQC Peer compare the accuracy and precision of analytical processes between laboratories and peer groups. This information can be extremely valuable, indicating the user’s performance relative to their peer group and also providing powerful troubleshooting tools when attempting to resolve potential problems. To participate in IAMQC Peer, each individual laboratory submits their individual results or summary statistics (mean, standard deviation, and number of data points) to the central database maintained by Technopath Manufacturing Ltd. Laboratories data may be submitted manually on-line or, alternatively, captured by one of our many interfacing options. The information provided by IAMQC Peer can be used on a monthly basis to evaluate how well lab’s methods are operating, relative to the overall peer group. Users can also look at this peer data in real-time, interactive, tables online, when they are investigating a potential problem with accuracy or precision for an individual method. -

 Precision. Confidence. Compliance. Technopath is proud to partner with Diagast, a global leader in immunohaematology, to deliver a comprehensive range of quality control solutions designed to ensure accuracy, reliability, and full regulatory compliance in transfusion laboratories with ISO 15189 requirements. Diagast’s immunohaematology quality control portfolio supports laboratories in maintaining the highest standards across blood grouping, antibody screening, crossmatching, and reagent performance verification—helping safeguard patient outcomes at every stage of the transfusion pathway. Comprehensive Quality Control for Transfusion Safety In immunohaematology, precision is critical. Diagast’s quality controls are specifically developed to:
Precision. Confidence. Compliance. Technopath is proud to partner with Diagast, a global leader in immunohaematology, to deliver a comprehensive range of quality control solutions designed to ensure accuracy, reliability, and full regulatory compliance in transfusion laboratories with ISO 15189 requirements. Diagast’s immunohaematology quality control portfolio supports laboratories in maintaining the highest standards across blood grouping, antibody screening, crossmatching, and reagent performance verification—helping safeguard patient outcomes at every stage of the transfusion pathway. Comprehensive Quality Control for Transfusion Safety In immunohaematology, precision is critical. Diagast’s quality controls are specifically developed to:- Monitor assay performance across routine and specialised testing
- Verify reagent integrity and system functionality
- Ensure compliance with ISO 15189 and local regulatory standards (HPRA, UKAS)
- Reduce risk of transfusion errors and improve laboratory confidence




