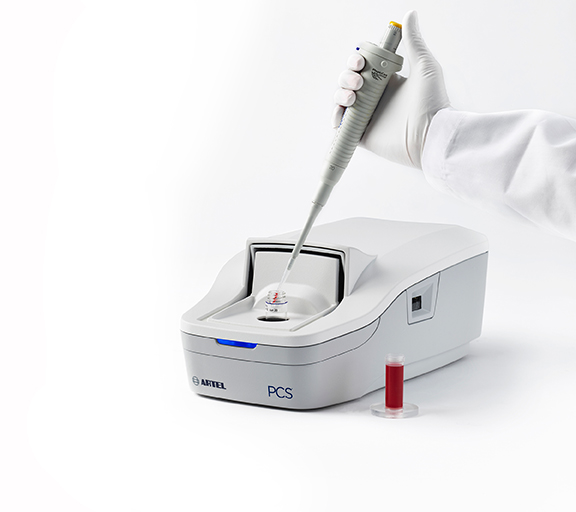
Features
Complete View
Intuitive dashboards provide a complete overview for managing and reviewing each pipette’s and operator’s status, from anywhere in your organisation.
Workflow
Email notifications for calibrations, competency assessments, and for overdue tasks. Review, approve and sign-off on calibration reports electronically. Generate calibration and inventory labels.
Pipette Inventory and QC
Maintain and manage your entire pipette inventory for complete QC of each device. Schedule calibrations and interim performance verification at defined intervals, and if required perform calibrations on an ad-hoc basis. Pipettes that have been calibrated outside of PCS Software can have those calibration records uploaded and become part of the pipette’s history.
Compliance
Facilitates compliance to 21 CFR Part 11. Allows user authentication and privilege management, versioning of calibration, training plans and electronic signatures. Provides a complete audit trail.
Testing Requirements
Customise tolerances based on your lab’s requirements to provide pass/fail results.
Operator Assessment
Schedule and document operator performance for internal and external regulatory compliance
Key Applications
Calibrate pipettes in the lab in which they are used; review results anywhere.
Standardize pipetting technique with scheduled and documented operator competency assessments.
Conduct interim performance verifications to supplement full calibration programs.
Verify pipette performance before and after maintenance, or following outsourced service.
Perform “quick checks” when pipetting performance or operator technique is in question.
Upload calibration certificates and repair documentation, as well as summary statistics for pipettes calibrated outside of PCS Software, including Artel Data Manager, Artel Pipette Tracker and third party or manufacturer services.